[ Home ] [ Controlled Substances ] [ Depressants ]
MIDAZOLAM
|
Midazolam injection is used to produce sleepiness or drowsiness and relieve anxiety before surgery or certain procedures. When midazolam is used before surgery, the patient will not remember some of the details about the procedure. Midazolam injection is also used as an anesthesia to produce loss of consciousness before and during surgery. Midazolam injection is also used to treat status epilepticus, a severe type of seizure where there is more than one seizure within 5 minutes. Midazolam is a benzodiazepine. Benzodiazepines belong to the group of medicines called central nervous system (CNS) depressants, which are medicines that slow down the nervous system. This medicine is given only by or under the direct supervision of your doctor.Pediatric:
Appropriate studies have not been performed on the relationship of age to the effects of midazolam in children below 6 months of age.Safety and efficacy have not been established.
Geriatric:
Appropriate studies have not been performed on the relationship of age to the effects of midazolam in the geriatric population.Safety and efficacy have not been established.
Other Interactions:
- Ethanol
- Grapefruit Juice
Other Medical Problems:
Make sure you tell your doctor if you have any other medical problems, especially:
- Apnea (temporary stopping of breathing)
- Heart disease
- Hypoventilation (slow breathing)
- Infections
- Lung disease, severe
- Lung or airway blockage- Use with caution. May increase risks for more serious side effects
- Congestive heart failure
- Kidney disease
- Liver disease - Use with caution. The effects may be increased because of slower removal of the medicine from the body
- Glaucoma, acute narrow-angle - Should not be used in patients with this condition
 Midazolam (Injection Route):
Midazolam (Injection Route):
https://www.mayoclinic.org/drugs-supplements/midazolam-injection-route/description/drg-20064813
 Midazolam (Oral Route) (Versed):
Midazolam (Oral Route) (Versed):
https://www.mayoclinic.org/drugs-supplements/midazolam-oral-route/description/drg-20072182
Prescribed for:
What Conditions does it Treat?
- Induce temporary amnesia
- Anxious
- Anxiety associated with an operation
- Additional medication for calming
- Calming of pediatric patient by sedative administration
- Sedation with ability to respond to stimulation or speech
- Additional agent to induce general anesthesia
Uses:
This medication is used in children before a procedure or anesthesia to cause drowsiness, decrease anxiety, and cause forgetfulness of the surgery or procedure.It should be used while the child is under the care of a health professional.
It is not for home or long-term use.Midazolam belongs to a class of medications called benzodiazepines, which produce a calming effect on the brain and nerves (central nervous system). It is thought to work by increasing the effect of a certain natural chemical (GABA) in the brain.
Before using:
Tell your doctor or pharmacist your medical history, especially of:Consult your doctor or pharmacist if you have:
- Kidney disease
- Liver disease
- Breathing problems (e.g., chronic obstructive pulmonary disease-COPD, sleep apnea)
- Heart disease (e.g., congestive heart failure)
- Glaucoma (open-angle)
- Personal or family history of a substance use disorder (such as overuse of or addiction to drugs/alcohol)
- glaucoma (narrow-angle)
Precautions:
- This drug may make you dizzy or drowsy
- Alcohol or marijuana (cannabis) can make you more dizzy or drowsy
- Do not drive, use machinery, or do anything that needs alertness until you can do it safely.
- Avoid alcoholic beverages.
- Talk to your doctor if you are using marijuana (cannabis).
WARNINGS:
Midazolam has rarely caused very serious breathing problems (such as rapid/slow/shallow breathing, trouble breathing), especially if used with other medications that cause drowsiness (including opioid medications such as morphine). This medication should be used only in a hospital or medical office under the care of a health professional.Read Reviews (8):
https://www.webmd.com/drugs/drugreview-16685-midazolam-oral.aspx?drugid=16685&drugname=midazolam-oral
 Midazolam HCL (Versed):
Midazolam HCL (Versed):
https://www.webmd.com/drugs/2/drug-16685/midazolam-oral/details
IMPORTANT WARNING:
Midazolam may cause serious or life-threatening breathing problems such as shallow, slowed, or temporarily stopped breathing. Your child should only receive this medication in a hospital or doctor's office that has the equipment that is needed to monitor his or her heart and lungs and to provide life-saving medical treatment quickly if his or her breathing slows or stops. Your child's doctor or nurse will watch your child closely after he or she receives this medication to make sure that he or she is breathing properly. Tell your child's doctor if your child has a severe infection or if he or she has or has ever had any airway or breathing problems or heart or lung disease. Tell your child's doctor and pharmacist if your child is taking any of the following medications: antidepressants; barbiturates such as secobarbital (Seconal); droperidol (Inapsine); medications for anxiety, mental illness, or seizures; narcotic medications for pain such as fentanyl (Actiq, Duragesic, Sublimaze, others), morphine (Avinza, Kadian, MS Contin, others), and meperidine (Demerol); sedatives; sleeping pills; or tranquilizers.
 Midazolam Injection:
Midazolam Injection:
https://medlineplus.gov/druginfo/meds/a609014.html
Dravet syndrome:
Midazolam is a benzodiazepine that is given to children before a medical procedure to relieve anxiety. It also is used as the first-line treatment of status epilepticus in children. Status epilepticus is when seizures last longer than five minutes, or multiple seizures occur close together without the patient recovering between them. Children with Dravet syndrome often experience status epilepticus, which can be life-threatening. Midazolam is a so-called rescue medicine that can be used to stop a seizure before it progresses to a medical emergency.
Dentistry (1993 Article):
Introduction: Much interest has been focused on the use of midazolam (Versed~Roche Laboratories of Hoffman LaRoche, Nutley, NJ) for conscious sedation in pediatric dentistry. The drug has been used as a preanesthetic sedative in adults and more recently in children. However, studies are lacking concerning its use in pediatric dentistry. The purpose of this paper is to review the pharmacokinetics of midazolam in children and its routes of administration including intravenous, oral rectal, and nasal routes.
Summary: Midazolam is a short-acting, water-soluble benzodiazepine. It has anxiolytic, sedative, hypnotic, anticonvulsant, muscle-relaxant, and anterograde amnesic effects. The drughas been used as a preanesthetic sedative in adults, andmore recently in children. This paper reviewed the pharmacokinetics of midazolam and its routes of administration in children. Intranasal administration was found tohave many advantages including rapid onset of sedation, ease of administration, and safety. The use of intranasal midazolam together with nitrous oxide/oxygen for conscious sedation of children during dental treatment should be investigated.


https://www.aapd.org/globalassets/media/publications/archives/kupietzky-15-04.pdf
Anesthesia:
A recent article from the California Society of Anesthesiologists BULLETIN underscored the danger of respiratory depression, morbidity, and mortality while using the remarkably effective and very useful drug, midazolam. This letter asks all anesthesiologists to be personally vigilant when using midazolam and to inform colleagues (endoscopist, dentists, etc.) concerning the appropriate dosage and potential hazards of midazolam.

Epilepsy:
Midazolam is a rescue medication that is approved for use:
- For short-term treatment of increased or frequent seizures called seizure clusters or acute repetitive seizures.
- In children over 12 years old and in adults.
- It is not supposed to be taken daily.
- This medicine does not take the place of your daily seizure medicine.
Nayzilam (midazolam) nasal spray Used to treat
Absence Seizures,
Atonic Seizures,
Atypical Absence Seizures,
Clonic Seizures,
Focal Impaired Awareness or Complex Partial Seizures,
Febrile Seizures,
Myoclonic Seizures,
Secondarily Generalized Seizures or Bilateral Tonic Clonic Seizure,
Focal Aware or Simple Partial Seizure,
Tonic Seizures, Tonic-clonic Seizures, Unknown Onset

https://www.epilepsy.com/medications/midazolam-nasal


https://www.ucb.com/_up/ucb_com_products/documents/Nayzilam%20COL%20Rev.%205-2019.pdf
Breastfeeding:Summary of Use During Lactation:
The small amounts of midazolam excreted into breastmilk would not be expected to cause adverse effects in most breastfed infants.Two expert panels advocates waiting for at least 4 hours after a single intravenous dose of midazolam (e.g., for endoscopy) before resuming nursing.
However, after a single dose of diazepam, as for sedation before a procedure or for a seizure, there is usually no need to wait to resume breastfeeding, although with a newborn or preterm infant, a cautious approach would be to wait a period of 6 to 8 hours before resuming nursing. After general anesthesia, breastfeeding can be resumed as soon as the mother has recovered sufficiently from general anesthesia to nurse. When a combination of anesthetic agents is used for a procedure, follow the recommendations for the most problematic medication used during the procedure.With prolonged use (days) of intravenous therapy, an active metabolite can accumulate in the mother and might affect the infant, but data in breastfeeding are lacking.
Drug Levels:
Midazolam is about 36% bioavailable orally in adults. It is metabolized to 1-hydroxymidazolam (60 to 70%) and 4-hydroxymidazolam (5%) which are about equipotent to midazolam. The half-life of 1-hydroxymidazolam is about 12 hours in adults and can accumulate with prolonged or repeated doses or in renal impairment.Alternate Drugs to Consider:
- (IV Sedation) Dexmedetomidine
- Etomidate
- Methohexital
- Propofol
- (Oral for Sleep) Zaleplon
- Zolpidem
 Modafinil Drug Levels and Effects:
Modafinil Drug Levels and Effects:
https://www.ncbi.nlm.nih.gov/books/NBK513058/
Versed Pharmacokinetics:
Absorption after I.M. administration appears to be 80% to 100% and after oral administration 40% to 50%.
Drug has a large volume of distribution and is about 97% protein-bound. Drug crosses the placental barrier and enters fetal circulation.
Metabolized in the liver.
Metabolites are excreted in urine. Half-life of drug is 1 1/4 to 12 1/3 hours. Half-life is prolonged in obese patients, the elderly, and seriously ill neonates.
 Midazolam hydrochloride Versed:
Midazolam hydrochloride Versed:
https://www.glowm.com/resources/glowm/cd/pages/drugs/m047.html
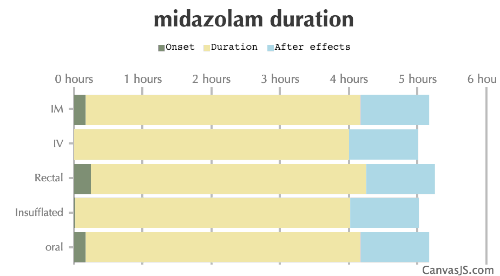
| Duration: A common hypnotic, sedative and anxiolytic benzodiazepine. High doses may cause amnesia and loss of inhibitions. Unusually, it is water soluble, and commonly used as a premedication for sedation as the solubility makes it better for IV use than other benzodiazepines. | |||
| Route | Onset | Duration | After Effects |
|---|---|---|---|
| Tripsit Factsheets | |||

Midazolam Basic Information: http://drugs.tripsit.me/midazolam | |||
| IM: | 10-20 minutes | 4-8 hours | 1-12 hours |
| IV: | 0-1 minutes | 4-8 hours | 1-12 hours |
| Rectal: | 15-25 minutes | 4-8 hours | 1-12 hours |
| Insufflated: | 1-5 minutes | 4-8 hours | 1-12 hours |
| Oral: | 10-40 minutes | 4-8 hours | 1-12 hours |
| |||
| Avoid: All other CNS depressants. | |||
| Effects: Anxiolytic, Sedative, Muscle Relaxant, Amnesia, Dystaxia, Hypnotic. | |||
Midazolam, previously marketed under the trade name Versed, is a medication used for anesthesia, procedural sedation, trouble sleeping, and severe agitation. Midazolam is a short-acting benzodiazepine central nervous system (CNS) depressant. Pharmacodynamic properties of midazolam and its metabolites, which are similar to those of other benzodiazepines, include sedative, anxiolytic, amnesic and hypnotic activities. Benzodiazepine pharmacologic effects appear to result from reversible interactions with the y-amino butyric acid (GABA) benzodiazepine receptor in the CNS, the major inhibitory neurotransmitter in the central nervous system. The action of midazolam is readily reversed by the benzodiazepine receptor antagonist, flumazenil. Data from published reports of studies in pediatric patients clearly demonstrate that oral midazolam provides safe and effective sedation and anxiolysis prior to surgical procedures that require anesthesia as well as before other procedures that require sedation but may not require anesthesia. The most commonly reported effective doses range from 0.25 to 1 mg/kg in children (6 months to <16 years). The single most commonly reported effective dose is 0.5 mg/kg. Time to onset of effect is most frequently reported as 10 to 20 minutes. The effects of midazolam on the CNS are dependent on the dose administered, the route of administration, and the presence or absence of other medications.
 Midazolam:
Midazolam:
https://drugs.ncats.io/drug/R60L0SM5BC
How Does Execution Drug Midazolam Work?
States can still use the sedative drug midazolam in lethal injections, according to today's Supreme Court decision. But how exactly does the drug work, and why do some say that it's unreliable? In a 5-to-4 vote, the court ruled that using midazolam does not violate the Eighth Amendment, which prohibits "cruel and unusual punishment." In executions, the drug has been used to induce unconsciousness before other drugs are administered to stop an inmate's breathing and stop the heart. However, midazolam was involved in several botched executions last year, including the case of Clayton Lockett from Oklahoma, who lived for about 45 minutes after he was administered drugs for lethal injection, and was seen convulsing and writhing before dying of a heart attack. (It was later determined that the IV line used to deliver the drugs in Lockett's execution was not properly placed.) In another controversial execution in Ohio, which also used midazolam, the inmate seemed unconscious at first, but then made loud snorting and choking noises before being pronounced dead, according to the New York Times. The combination of drugs used in the Ohio case had not previously been used in an execution.
- https://www.livescience.com/51384-execution-drug-midazolam-effect.html

https://erowid.org/experiences/subs/exp_Pharms_Midazolam.shtml

https://www.reddit.com/search/?q=midazolam
| Maximum Dosage: | |
|---|---|
| Prescribers Digital Reference | |

Midazolam hydrochloride Drug Summary: https://www.pdr.net/drug-summary/Midazolam-Hydrochloride-Syrup-midazolam-hydrochloride-1837 | |
| Adults: | 1 spray (5 mg)/dose intranasal up to 2 doses/episode for acute repetitive seizures. Specific maximum dosage information for other routes or indications is not available; the dose required is dependent on route of administration, indication for therapy, and clinical response. |
| Geriatric: | 1 spray (5 mg)/dose intranasal up to 2 doses/episode for acute repetitive seizures. Specific maximum dosage information for other routes or indications is not available; the dose required is dependent on route of administration, indication for therapy, and clinical response. |
| Adolescents: | 1 mg/kg PO (Max: 20 mg/dose) for procedural sedation; a total dose up to 5 mg IV may be necessary for amnesia induction; 1 spray (5 mg)/dose intranasal up to 2 doses/episode for acute repetitive seizures; specific maximum dosage information for other routes or indications is not available; the dose required is dependent on route of administration, indication, and clinical response. |
| Children: | 12 years: 1 mg/kg PO (Max: 20 mg/dose) as a single dose for procedural sedation; a total dose up to 0.4 mg/kg IV (Usual Max: 10 mg) may be necessary for amnesia induction; 1 spray (5 mg)/dose intranasal up to 2 doses/episode for acute repetitive seizures; specific maximum dosage information for other routes or indications is not available; the dose required is dependent on route of administration, indication, and clinical response. |
| Children: | 6 to 11 years: 1 mg/kg PO (Max: 20 mg/dose) as a single dose for procedural sedation; a total dose up to 0.4 mg/kg IV (Usual Max: 10 mg) may be necessary for amnesia induction; specific maximum dosage information for other routes or indications is not available; the dose required is dependent on route of administration, indication, and clinical response. |
| Children: | 1 to 5 years: 1 mg/kg PO (Max: 20 mg/dose) as a single dose for procedural sedation; a total dose up to 0.6 mg/kg IV (Usual Max: 6 mg) may be necessary for amnesia induction; specific maximum dosage information for other routes or indications is not available; the dose required is dependent on route of administration, indication, and clinical response. |
| Infants: | 6 to 11 months: 1 mg/kg PO (Max: 20 mg/dose) as a single dose for procedural sedation; a total dose up to 0.6 mg/kg IV (Usual Max: 6 mg) may be necessary for amnesia induction; specific maximum dosage information for other routes or indications is not available; the dose required is dependent on route of administration, indication, and clinical response. |
| Infants: | 1 to 6 months: Specific maximum dosage information is not available; the dose required is dependent on route of administration, indication, and clinical response. |
| Neonates: | Specific maximum dosage information is not available; the dose required is dependent on route of administration, indication, and clinical response. |
| Black Box Warnings: |
|---|

Midazolam (Rx) Brand and Other Names: Seizalam, Versed (DSC): https://reference.medscape.com/drug/seizalam-versed-dsc-midazolam-342907 |
|
Respiratory depression/arrest has been associated with use, especially when used for sedation in noncritical care settings Use lower end of dosing range in debilitated patients, including the elderly Do not administer by rapid IV injection in neonates (hypotension and seizures reported, especially when used concomitantly with fentanyl) Respiratory depression, airway obstruction, desaturation, hypoxia, and apnea, particularly when used with concomitant CNS depressants (eg, opioids), have been reported Should be used only in settings (eg, hospital, ambulatory care settings, including physicians' or dentists' offices) that can provide continuous monitoring of respiratory and cardiac function; immediate availability of resuscitative drugs and age- and size-appropriate equipment for ventilation and intubations, as well as personnel trained in their use and skilled in airway management, should be ensured For deeply sedated patients, a dedicated individual other than the practitioner performing the procedure should monitor the patient throughout the procedure Use of the 1 mg/mL formulation or dilution of the 1 mg/mL or 5 mg/mL formulation is recommended to facilitate slower injection Adult dosing
Pediatric dosing
Neonates
General anesthetics and sedation drugs in young children and pregnant women
|
| Side Effects: |
|---|
| RxList |
midazolam injection (midazolam) drug: https://www.rxlist.com/midazolam-injection-drug.htm |
Common side effects of Midazolam include:
|
Liver:
Midazolam therapy has not been associated with serum aminotransferase elevations and has not been linked to cases of clinically apparent liver injury.
Midazolam Hepatotoxicity:
Midazolam, like other benzodiazepines, is rarely associated with serum ALT elevations. Clinically apparent liver injury from midazolam has not been reported and must be extremely rare, if it occurs at all. Cases of clinically apparent liver injury have been reported with other benzodiazepines including alprazolam, chlordiazepoxide, clonazepam, diazepam, flurazepam and triazolam. The clinical pattern of acute liver injury from benzodiazepines is typically cholestatic, but hepatocellular patterns of injury have been reported with chlorazepate and clotiazepam. The injury is usually mild to moderate in severity with a time to onset of 1 to 6 months. Fever and rash are uncommon as is autoantibody formation.E Likelihood score: E (unlikely cause of clinically apparent liver injury).
 Midazolam Overview:
Midazolam Overview:
https://www.ncbi.nlm.nih.gov/books/NBK548691/
Important Information:
Midazolam can slow or stop your breathing, especially if you have recently used a narcotic (opioid) medication. Midazolam is given in a hospital, dentist office, or other clinic setting where your vital signs can be watched closely.
You should not take midazolam if you have narrow-angle glaucoma, if you are allergic to cherries, or if you are allergic to midazolam or similar medicines (Valium, Xanax, Ativan, and others).Do not drink alcohol for at least 24 hours after taking midazolam.
Do not drink alcohol for at least 24 hours after taking midazolam. This medication can increase the effects of alcohol, which could be dangerous.Grapefruit and grapefruit juice may interact with midazolam and lead to potentially dangerous effects. Avoid the use of grapefruit products while taking midazolam.
Midazolam injection can cause extreme drowsiness that may last for 24 hours after you have received the medication. Older adults may feel sleepy for even longer.Avoid driving or doing anything that requires you to be awake and alert until the effects of midazolam have worn off completely.
Interactions:
Drug Interactions (461) Alcohol/Food Interactions (2) Disease Interactions (12)
What other drugs will affect Midazolam?
Shortly after you take midazolam, taking other drugs that make you sleepy or slow your breathing can cause dangerous side effects. Tell your doctor if you regularly use a sleeping pill, narcotic pain medicine, prescription cough medicine, a muscle relaxer, or medicine for anxiety, depression, or seizures. Many drugs can interact with midazolam. Not all possible interactions are listed here. Tell your doctor about all your current medicines, especially:This list is not complete and many other drugs can interact with midazolam. This includes prescription and over-the-counter medicines, vitamins, and herbal products. Give a list of all your medicines to any healthcare provider who treats you.
- antifungal medicine
- an antibiotic
- an antidepressant
- heart or blood pressure medicine
- antiviral medicine to treat hepatitis C or HIV/AIDS
- seizure medication
- tuberculosis medication
A total of 461 drugs are known to interact with Midazolam.
- 31 major drug interactions
- 391 moderate drug interactions
- 39 minor drug interactions
 Midazolam Oral Versed:
Midazolam Oral Versed:
https://www.drugs.com/mtm/midazolam.html

https://www.uptodate.com/contents/midazolam-drug-information?source=see_link

https://www.medicines.org.uk/emc/product/6420/smpc

https://www.pfizer.com/products/product-detail/midazolam


https://www.pfizer.ca/sites/default/files/201712/2017.10.25_Midazolam_PM_PF_E_210318.pdf


https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/208878Orig1s000lbl.pdf
History:
Midazolam is among about 35 benzodiazepines currently used medically, and was synthesized in 1975 by Walser and Fryer at Hoffmann-LaRoche, Inc in the United States. Owing to its water solubility, it was found to be less likely to cause thrombophlebitis than similar drugs. The anticonvulsant properties of midazolam were studied in the late 1970s, but not until the 1990s did it emerge as an effective treatment for convulsive status epilepticus. As of 2010, it is the most commonly used benzodiazepine in anesthetic medicine. In acute medicine, midazolam has become more popular than other benzodiazepines, such as lorazepam and diazepam, because it is shorter lasting, is more potent, and causes less pain at the injection site. Midazolam is also becoming increasingly popular in veterinary medicine due to its water solubility.
Midazolam can be given by mouth, intravenously, or injection into a muscle, by spraying into the nose, or through the cheek. When given intravenously, it typically begins working within five minutes; when injected into a muscle, it can take fifteen minutes to begin working. Effects last for between one and six hours. Side effects can include a decrease in efforts to breathe, low blood pressure, and sleepiness. Tolerance to its effects and withdrawal syndrome may occur following long-term use. Paradoxical effects, such as increased activity, can occur especially in children and older people. There is evidence of risk when used during pregnancy but no evidence of harm with a single dose during breastfeeding. Midazolam was patented in 1974 and came into medical use in 1982.
How does execution drug midazolam work? - States can still use the sedative drug midazolam in lethal injections, according to today's Supreme Court decision. But how exactly does the drug work, and why do some say that it's unreliable? In a 5 ... Tuesday June 30, 2015 - foxnews.com A look at Midazolam, the controversial drug at the center of Arkansas execution scandal - Arkansas has been at the center of a controversial legal battle because of its plan to try and speed up inmate executions before its supply of the drug midazolam expires. The sedative, one of the ... What Is Midazolam, the Lethal Injection Drug Approved by the Supreme Court - See more of our trusted coverage when you search. Prefer Newsweek on Google to see more of our trusted coverage when you search. The Supreme Court in a 5-4 decision on Monday upheld the use of ... Supreme Court to review use of midazolam as execution drug - WASHINGTON (AP) - The Supreme Court is stepping into the issue of lethal injection executions for the first time since 2008 in an appeal filed by death row inmates in Oklahoma. The justices agreed ... Is Execution Drug Midazolam Constitutional? Supreme Court Debates Challenge to Lethal Injection Drug - Supreme Court justices engaged in an impassioned debate Wednesday about capital punishment — part of arguments over a single drug that has been used in several botched executions. The justices heard ... Midazolam - Uses, Side Effects, and More - What is midazolam used for? Midazolam is commonly used to treat the following conditions. To relax you and make you sleepy before a surgery or medical procedure. Certain type of seizure disorders ... Can Midazolam Drug Be an Effective Treatment Option for Seizures During Emergency? - Status Epilepticus is a type of epilepsy which is characterized by seizures followed by one after the other, without the recovery of consciousness. Patients who experience seizures outside the ... Midazolam : Interaction with Foods, Herbs & Alcohol - Are you taking Midazolam? Are you aware of the dangers of combining Midazolam with certain foods, herbs, or alcohol? Drug-food-interactions are seldom discussed but can lead to undesirable side ... Major shortage of vital epilepsy drug taken by thousands – as man dies after being left without medication - HUNDREDS of thousands of Brits could be left without a life-saving epilepsy drug as the NHS faces a shortage of vital seizure medication. The shortage comes as a coroner blamed a shortage of ... Supreme Court will review use of midazolam as execution drug - WASHINGTON – The Supreme Court is stepping into the issue of lethal injection executions for the first time since 2008 in an appeal filed by death row inmates in Oklahoma. The justices agreed Friday ...
| ||
| Depressants | Link to this page |