[ Home ] [ Controlled Substances ] [ Depressants ]
FLURAZEPAM
|
Dalmane
Flurazepam (known under the brand names Dalmane and Dalmadorm) is a drug which is a benzodiazepine derivative. It is a hypnotic agent which does not appear to decrease dream time as measured by rapid eye movements (REM). Furthermore, it decreases sleep latency and number of awakenings for a consequent increase in total sleep time. Flurazepam binds to an allosteric site on GABA-A receptors. Binding potentiates the action of GABA on GABA-A receptors by opening the chloride channel within the receptor, causing chloride influx and hyperpolarization. Flurazepam is useful for the treatment of insomnia characterized by difficulty in falling asleep, frequent nocturnal awakenings, and/or early morning awakening. Flurazepam can be used effectively in patients with recurring insomnia or poor sleeping habits, and in acute or chronic medical situations requiring restful sleep.
 Flurazepam:
Flurazepam:
https://drugs.ncats.io/drug/IHP475989U
| Flurazepam Hydrochloride Capsules: Are a hypnotic agent useful for the treatment of insomnia characterized by difficulty in falling asleep, frequent nocturnal awakenings, and/or early morning awakening. Flurazepam Hydrochloride Capsules can be used effectively in patients with recurring insomnia or poor sleeping habits, and in acute or chronic medical situations requiring restful sleep. Sleep laboratory studies have objectively determined that Flurazepam Hydrochloride Capsules are effective for at least 28 consecutive nights of drug administration. Since insomnia is often transient and intermittent short-term use is usually sufficient. Prolonged use of hypnotics is usually not indicated and should only be undertaken concomitantly with appropriate evaluation of the patient. |
| RxList |
|---|
Excerpt from rxlist.com: https://www.rxlist.com/flurazepam-hydrochloride-drug.htm |
This medication is used to treat a certain sleep problem (insomnia). It may help you fall asleep faster, stay asleep longer, and lessen how often you wake up during the night, so you can get a better night's rest. Flurazepam belongs to a class of drugs called sedative-hypnotics. It acts on your brain to produce a calming effect. Use of this medication is usually limited to treatment periods of 1 to 2 weeks or less. If your insomnia continues for a longer time, talk to your doctor to see if you need other treatment. Although unlikely, this drug can rarely cause temporary short-term memory loss. Though it helps many people, this medication may sometimes cause addiction. This medication may cause withdrawal reactions, especially if it has been used regularly for a long time or in high doses.
 Excerpt from Webmd:
Excerpt from Webmd:
https://www.webmd.com/drugs/2/drug-7409/dalmane-oral/details
Class Name: Sedative/Hypnotics
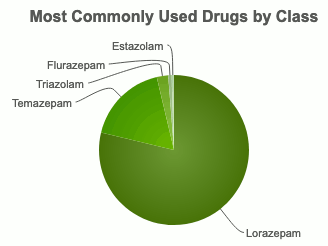
ESTAZOLAM | FLURAZEPAM | TRIAZOLAM | TEMAZEPAM | LORAZEPAM
| Duration: Benzodiazepine with an extremely long half life, between 40 and 250 hours. Mainly used for general anxiety disorder. Sedating, hypnotic, anxiolytic. Potential for amnesia and reduced inhibitions in overdose. | |||
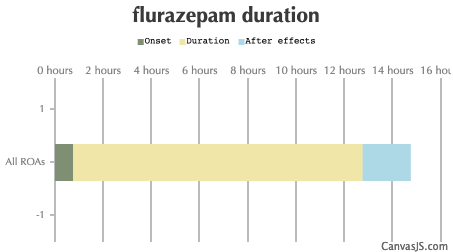
| Route | Onset | Duration | After Effects |
|---|---|---|---|
| Tripsit Factsheets | |||

Flurazepam Basic Information: http://drugs.tripsit.me/flurazepam | |||
| All ROAs: | 45-90 minutes | 12-16 hours | 2-48 hours |
| |||
| Avoid: All other CNS depressants. | |||
| Effects: Anxiolytic, Sedative, Muscle Relaxant, Amnesia, Dystaxia, Hypnotic. | |||
Important Information:
Some people using flurazepam have engaged in activity such as driving, eating, walking, making phone calls, or having sex and later having no memory of the activity.
This medication may impair your thinking or reactions. Be careful if you drive or do anything that requires you to be alert.
Do not drink alcohol. Dangerous side effects can occur when alcohol is combined with flurazepam.
Interactions:
Drug Interactions (418) Alcohol/Food Interactions (2) Disease Interactions (9)
What other drugs will affect Flurazepam?
Taking flurazepam with other drugs that make you sleepy or slow your breathing can cause dangerous side effects or death. Ask your doctor before taking a sleeping pill, narcotic pain medicine, prescription cough medicine, a muscle relaxer, or medicine for anxiety, depression, or seizures. Other drugs may interact with flurazepam, including prescription and over-the-counter medicines, vitamins, and herbal products. Tell each of your health care providers about all medicines you use now and any medicine you start or stop using.A total of 418 drugs are known to interact with Flurazepam.
- 26 major drug interactions
- 359 moderate drug interactions
- 33 minor drug interactions
 Flurazepam Drug Information:
Flurazepam Drug Information:
https://www.drugs.com/mtm/flurazepam.html
 User Reviews from Drugs.com:
User Reviews from Drugs.com:
https://www.drugs.com/comments/flurazepam/dalmane.html

https://dailymed.nlm.nih.gov/dailymed/medguide.cfm?setid=3839ca57-1c3d-4fb3-a00a-6569b2b4447f
| Maximum Dosage: | |
|---|---|
| Prescribers Digital Reference | |

Prescribers Digital Reference Flurazepam Drug Summary : https://www.pdr.net/drug-summary/Flurazepam-Hydrochloride-flurazepam-hydrochloride-1969 | |
| Adults: | 30 mg/day PO at bedtime. |
| Elderly | 15 mg/day PO at bedtime. |
| Adolescents: | >= 15 years: 30 mg/day PO at bedtime. |
| Adolescents: | < 15 years: Safety and efficacy have not been established. |
| Children: | Safety and efficacy have not been established. |
| Black Box Warnings: |
|---|
|
Concomitant use of benzodiazepines and opioids may result in profound respiratory depression, coma, and death; administer concomitantly when there are no alternative options; limit dosages and durations to minimum required; monitor for signs and symptoms of respiratory depression and sedation Addiction, abuse, and misuse
|
| Side Effects: |
| Get emergency medical help if you have signs of an allergic reaction: hives; nausea and vomiting; difficulty breathing; swelling of your face, lips, tongue, or throat. |
|---|
| RxList |
Generic Name: flurazepam; Brand Name: Dalmane: https://www.rxlist.com/dalmane-drug/patient-images-side-effects.htm |
Call your doctor at once if you have:
|
| Some people using this medication have engaged in activity such as driving, eating, or making phone calls and later having no memory of the activity. If this happens to you, stop taking flurazepam and talk with your doctor. |
Common side effects may include:
|
| This is not a complete list of side effects and others may occur. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
Pediatric:
Appropriate studies have not been performed on the relationship of age to the effects of flurazepam in the pediatric population.Safety and efficacy have not been established.
Geriatric:
Appropriate studies performed to date have not demonstrated geriatric-specific problems that would limit the usefulness of flurazepam in the elderly. However, elderly patients are more likely to have unwanted effects (eg, severe drowsiness, dizziness, confusion, clumsiness, or unsteadiness) and age-related kidney, liver, or heart problems, which may require an adjustment in the dose for patients receiving flurazepam.Other Medical Problems:
Make sure you tell your doctor if you have any other medical problems, especially:
- Alcohol abuse, or history of
- Breathing problems or lung disease
- Depression, or history of
- Drug abuse or dependence, or history of
- Mental health problems, or history of - Use with caution. May make these conditions worse
- Kidney disease
- Liver disease - Use with caution. The effects may be increased because of slower removal of the medicine from the body
 Flurazepam (Oral Route) (Dalmane):
Flurazepam (Oral Route) (Dalmane):
https://www.mayoclinic.org/drugs-supplements/flurazepam-oral-route/description/drg-20072260
Liver:
As with most benzodiazepines, flurazepam therapy has not been associated with serum aminotransferase or alkaline phosphatase elevations; clinically apparent liver injury from flurazepam has been reported, but is very rare.
Flurazepam Hepatotoxicity:
Flurazepam, as with other benzodiazepines, is rarely associated with serum ALT elevations, and clinically apparent liver injury is rare. Only a few case reports of acute liver injury from flurazepam have been published and mostly before 1980. The latency to onset of acute liver injury has varied from a 2 to 6 months and the pattern of liver enzyme elevations was cholestatic. The injury is usually mild to moderate in severity and self-limited. Fever and rash have not been described nor has autoantibody formation.D Likelihood score: D (Possible rare cause of clinically apparent liver injury).
 Flurazepam Overview:
Flurazepam Overview:
https://www.ncbi.nlm.nih.gov/books/NBK548683/
Breastfeeding:Summary of Use During Lactation:
No information is available on the excretion of flurazepam into breastmilk. One case of infant sedation was reported in a woman taking flurazepam along with other sedating drugs during breastfeeding.Because of the long duration of action of flurazepam, an alternate hypnotic is preferred, especially while nursing a newborn or preterm infant.
Alternate Drugs to Consider:
 Flurazepam Drug Levels and Effects:
Flurazepam Drug Levels and Effects:
https://www.ncbi.nlm.nih.gov/books/NBK501727/


https://www.accessdata.fda.gov/drugsatfda_docs/label/2007/016721s076lbl.pdf

https://erowid.org/experiences/subs/exp_Pharms_Flurazepam.shtml
 Dalmane Image:
Dalmane Image:
https://commons.wikimedia.org/wiki/File:Dalmane15mg.jpgFlurazepam produces a metabolite with a long half-life, which may stay in the bloodstream for days. That is the reason why users experience a "hangover" effect the day after it was used. Intermediate half-life benzodiazepines are also useful for patients with difficulty in maintaining sleep (e.g. loprazolam, lormetazepam, temazepam)
Flurazepam was patented in 1968 and came into medical use the same year. Flurazepam, developed by Roche Pharmaceuticals was one of the first benzo hypnotics (sleeping pills) to be marketed. Flurazepam has abuse potential and should never be used with alcoholic beverages or any other substance that can cause drowsiness. Addictive and possibly fatal results may occur.
Flurazepam - Uses, Side Effects, and More - What is flurazepam used for? Flurazepam is commonly used to treat certain types of a sleep problem called insomnia, which can include symptoms like trouble falling asleep and waking up often during ... Friday July 26, 2024 - webmd.com Zomnia (Flurazepam) Drug Price and Information - Flurazepam (Zomnia) is a benzodiazepine, prescribed for sleep disorder. It works by slowing activity in the brain to allow sleep. The information provided on this page is intended to serve as a ... Nindral (15 mg) (Flurazepam) Drug Price and Information - The Price section compares costs of the same generic drugs across brands and is purely for information purpose. Medindia neither buys nor sells drugs. Flurazepam (Nindral (15 mg)) is a benzodiazepine, ...
| ||
| Depressants | Link to this page |







