[ Home ] [ Controlled Substances ] [ Opioids ]
TRAMADOL
|
About Tramadol:
- Tramadol is a strong painkiller. It's used to treat moderate to severe pain, for example after an operation or a serious injury.
- It's also used to treat long-standing pain when weaker painkillers no longer work.
- Tramadol is available only on prescription. It comes as tablets, capsules and liquid drops that you swallow. It can also be given by injection but this is usually only done in hospital.
Key Facts:
- Tramadol works by blocking pain signals from travelling along the nerves to the brain.
- The most common side effects of tramadol are feeling sick and dizzy.
- It's possible to become addicted to tramadol, but this is rare if you're taking it to relieve pain and your doctor is reviewing your treatment regularly.
- It's best not to drink alcohol with tramadol as you're more likely to get side effects like feeling sleepy.
- Tramadol is also called by the brand names:
- Invodol
- Larapam
- Mabron
- Maneo
- Marol
- Maxitram
- Oldaram
- Tilodol
- Tradorec
- Tramquel
- Tramulief
- Zamadol
- Zeridame
- Zydol
- Tramadol can be taken by adults and children aged 12 and over.
- The dose can vary but you should not normally take more than 400mg a day.
Tramadol (sold under the brand name Ultram) is a narcotic analgesic proposed for moderate to severe pain. Tramadol and its O-desmethyl metabolite (M1) are selective, weak OP3-receptor agonists. Opiate receptors are coupled with G-protein receptors and function as both positive and negative regulators of synaptic transmission via G-proteins that activate effector proteins. As the effector system is adenylate cyclase and cAMP located at the inner surface of the plasma membrane, opioids decrease intracellular cAMP by inhibiting adenylate cyclase. Subsequently, the release of nociceptive neurotransmitters such as substance P, GABA, dopamine, acetylcholine, and noradrenaline is inhibited. The analgesic properties of Tramadol can be attributed to norepinephrine and serotonin reuptake blockade in the CNS, which inhibits pain transmission in the spinal cord. The (+) enantiomer has the higher affinity for the OP3 receptor and preferentially inhibits serotonin uptake and enhances serotonin release. The (-) enantiomer preferentially inhibits norepinephrine reuptake by stimulating alpha(2)-adrenergic receptors. Tramadol is used primarily to treat mild-severe pain, both acute and chronic. Its analgesic effects take about one hour to come into effect and 2 h to 4 h to peak after oral administration with an immediate-release formulation. On a dose-by-dose basis, tramadol has about one-tenth the potency of morphine and is approximately equally potent when compared to pethidine and codeine. The most common adverse effects of tramadol include nausea, dizziness, dry mouth, indigestion, abdominal pain, vertigo, vomiting, constipation, drowsiness, and headache. Compared to other opioids, respiratory depression and constipation are considered less of a problem with tramadol.
 Tramadol:
Tramadol:
https://drugs.ncats.io/drug/39J1LGJ30J
Uses:
This medication is used to help relieve moderate to moderately severe pain. Tramadol is similar to opioid analgesics.It works in the brain to change how your body feels and responds to pain.
Used for:
Pain, chronic pain, neuropathic pain disorder characterized by stifftender & painful muscles
Before Using:
Tell your doctor or pharmacist your medical history, especially of:
- brain disorders (such as head injury, tumor, seizures)
- breathing problems (such as asthma, sleep apnea, chronic obstructive pulmonary disease-COPD)
- kidney disease
- liver disease
- mental/mood disorders (such as confusion, depression, suicidal thoughts)
- personal or family history of a substance use disorder (such as overuse of or addiction to drugs/alcohol)
- stomach/intestinal problems (such as blockage, constipation, diarrhea due to infection, paralytic ileus)
- difficulty urinating (such as due to enlarged prostate)
- gallbladder disease
- disease of the pancreas (pancreatitis)
- obesity
Precautions:
This drug may make you dizzy or drowsy.
Alcohol or marijuana (cannabis) can make you more dizzy or drowsy.
Do not drive, use machinery, or do anything that needs alertness until you can do it safely.
Avoid alcoholic beverages.
Talk to your doctor if you are using marijuana (cannabis).Tramadol may cause a condition that affects the heart rhythm (QT prolongation). QT prolongation can rarely cause serious (rarely fatal) fast/irregular heartbeat and other symptoms (such as severe dizziness, fainting) that need medical attention right away. The risk of QT prolongation may be increased if you have certain medical conditions or are taking other drugs that may cause QT prolongation.
Low levels of potassium or magnesium in the blood may also increase your risk of QT prolongation. This risk may increase if you use certain drugs (such as diuretics/"water pills") or if you have conditions such as severe sweating, diarrhea, or vomiting. Talk to your doctor about using tramadol safely.WARNINGS:
Tramadol has a risk for abuse and addiction, which can lead to overdose and death. Tramadol may also cause severe, possibly fatal, breathing problems. To lower your risk, your doctor should have you take the smallest dose of tramadol that works, and take it for the shortest possible time.
The risk for severe breathing problems is higher when you start this medication and after a dose increase, or if you take the wrong dose/strength.
Taking this medication with alcohol or other drugs that can cause drowsiness or breathing problems may cause very serious side effects, including death.
Other medications can affect the removal of tramadol from your body, which may affect how tramadol works.
Be sure you know how to take tramadol and what other drugs you should avoid taking with it. Get medical help right away if any of these very serious side effects occur: slow/shallow breathing, unusual lightheadedness, severe drowsiness/dizziness, difficulty waking up.User Reviews:
2937 Total User Reviews Tramadol Oral Read Reviews Condition: Pain (1683 Reviews): Effectiveness (3.22) Ease of Use (4.16) Satisfaction (2.98)
 Tramadol HCL (Ultram):
Tramadol HCL (Ultram):
https://www.webmd.com/drugs/2/drug-4398-5239/tramadol-oral/tramadol-oral/details
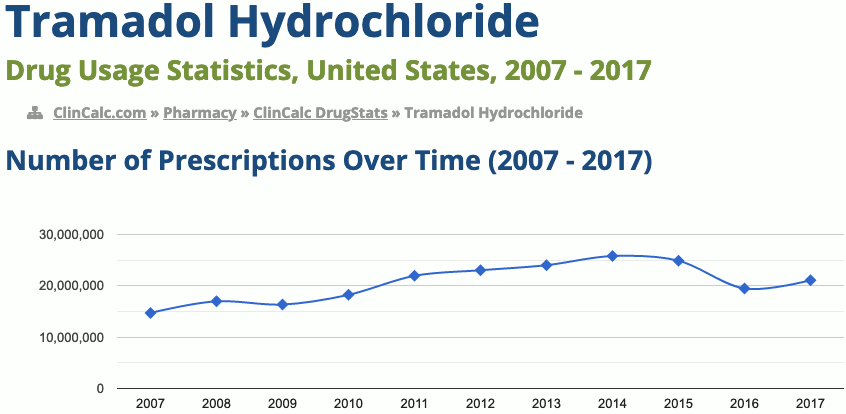
32nd most prescribed medicine in the United States for 2017

https://clincalc.com/DrugStats/Drugs/TramadolHydrochloride
WHO Report:Substance identification:
Tramadol is a white, bitter, crystalline and odourless powder soluble in water and ethanol. Tramadol is marketed as the hydrochloride salt and is available in a variety of pharmaceutical formulations for oral(tablets, capsules), sublingual (drops), intranasal, rectal (suppositories), intravenous, subcutaneous, and intramuscular administration. It is also available in combination with acetaminophen (paracetamol). Preparations of tramadol are available as immediate-and extended-release formulations.Effects on Central Nervous System:
Tramadol has been detected in a number of deaths. It is often present along with other drugs, including opioids, benzodiazepines and antidepressants,but fatalities have also been reported due to tramadol alone.Dependence Potential:
Evidence suggests that the development of physical dependence to tramadol is dose-related, and administration of supra-therapeutic doses leads to a similar dependence profile to morphine and other opioids such as oxycodone and methadone. There are reports of considerable number of people with tramadol dependence seeking help. Withdrawal symptoms include those typical of opioids such as pain, sweating, diarrhoea and insomnia as well as symptoms not normally seen with opioids and related to noradrenergic and serotonergic activity, such as hallucinations, paranoia, confusion and sensory abnormalities. Low dose tramadol use over extended periods is associated with a lower risk of dependence.Consistent with its opioid mechanism of action, human brain imaging has shown that tramadol activates brain reward pathways associated with abuse. While reports from people administered tramadol in controlled settings have shown that it is identified as opioid-like and tramadol has reinforcing effects in experienced opioid users, these effects may be weaker than those produced by opioids such as morphine and may be partially offset by unpleasant effects of tramadol such as sweating, tremor, agitation, anxiety and insomnia. The oral route of administration has been the predominant mode of tramadol abuse as it results in a greater opioid effect compared to other routes.It is unlikely that tramadol will be injected to any significant extent.
Recommendation:
The Committee was concerned by the increasing evidence for tramadol abuse in a number of countries in diverse regions, in particular the widespread abuse of tramadol in many low to middle income countries. Equally concerning was the clear lack of alternative analgesics for moderate to severe pain for which tramadol is used.

 (PDF page 20 / 22 pages) (41st Expert Committee on Drug Dependence):
(PDF page 20 / 22 pages) (41st Expert Committee on Drug Dependence):
https://www.who.int/medicines/access/controlled-substances/Annex_1_41_ECDD_recommendations_NPS_and_medicines_22012019.pdf#page=20
Liver:
Tramadol overdose can cause acute liver failure. Pharmacologic use of tramadol has not been associated with cases of clinically apparent drug induced liver disease.
Tramadol Hepatotoxicity:
Serum aminotransferase levels can be elevated in a small proportion of patients receiving tramadol, particularly with high doses. Intentional and accidental overdoses of tramadol can cause respiratory arrest as well as acute liver failure, several fatal instances of which have been reported. In these cases, however, the liver injury may have been caused by shock, hypoxia or ischemia secondary to the respiratory arrest. Liver injury attributed to tramadol overdose has also been associated with hyperammonemia, lactic acidosis and hepatic steatosis, suggestive of direct mitochondrial injury. In some situations, acute liver failure after tramadol overdose may be related to acetaminophen taken separately or in combination with tramadol. Clinically apparent idiosyncratic liver injury with recommended doses of tramadol has not been reported and must be rare, if it occurs at all.E Likelihood score: E (unlikely cause of clinically apparent liver injury).
 Tramadol Overview:
Tramadol Overview:
https://www.ncbi.nlm.nih.gov/books/NBK548235/
Harm reduction advice:
If you choose to use tramadol, taking some simple steps can reduce some of the risks and help you stay safer:
- Stay within recommended doses. The majority of serious side-effects from tramadol occur when using more than the recommended daily dosage. However, tramadol overdose can occur even from taking smaller doses, often in combination with other drugs.
- Don't use alone. Serious side effects such as seizures and low blood sugar can occur in certain people. Try to ensure that other people are with you when using tramadol, especially if it's your first time using.
- Avoid mixing drugs. Try to avoid mixing tramadol with other drugs, including prescription medication. Fatal overdose on tramadol alone is possible but is much more common when tramadol is mixed with other drugs, especially other central nervous system depressants. Mixing tramadol with prescribed medications can also lead to a range of dangerous and life-threatening conditions.
- Avoid injecting tablets and capsules. Injecting this form of tramadol whether IV, IM or subcutaneous risks overdose and death, and the fillers and binders in tramadol tablets when injected can cause local tissue necrosis, infection, pulmonary granulomas and increased risk of endocarditis and valvular heart injury.

http://thedrugswheel.com/?page=tramadol
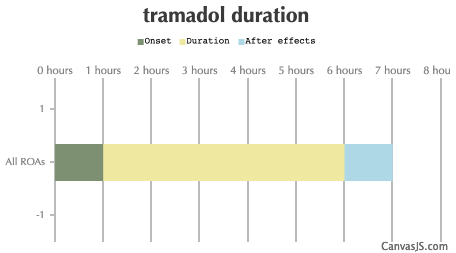
| Duration: A synthetic opioid analgesic, tramadol is used to treat moderate pain and can be considered a medium-strength opioid. Tramadol also has the unusual effect of being a serotonin releasing agent and a serotonin reuptake inhibitor, and as a consequence should not be taken in excess due to the risk of serotonin syndrome. Risk of seizures above 300mg doses. NOTE: Tramadol has a ceiling dose, where recreational effects are not increased. Risk of seizure at doses over 300mg. Tramadol decreases the seizure threshold, use extreme caution. Dosing insufflated is ineffective. | |||
| Route | Onset | Duration | After Effects |
|---|---|---|---|
| Tripsit Factsheets | |||

Tramadol Basic Information: http://drugs.tripsit.me/tramadol | |||
| All ROAs: | 1 hour | 5-7 hours | 1-12 hours |
| |||
| Effects: euphoria, elevated mood, overall feeling of contentedness, itching, drowsiness, vomiting, urinary retention, nausea, constipation, risk of seizure above 300mg | |||
| Avoid: benzos, alcohol, and other opiates | |||

https://erowid.org/experiences/subs/exp_Pharms_Tramadol.shtml

https://drugs-forum.com/forums/tramadol.399/
| Side Effects: |
| Get emergency medical help if you have signs of an allergic reaction (hives, difficult breathing, swelling in your face or throat) or a severe skin reaction (fever, sore throat, burning in your eyes, skin pain, red or purple skin rash that spreads and causes blistering and peeling). |
|---|
| RxList |
Ultram (tramadol hcl): https://www.rxlist.com/ultram-drug/patient-images-side-effects.htm |
| This medicine can slow or stop your breathing, and death may occur. A person caring for you should seek emergency medical attention if you have slow breathing with long pauses, blue colored lips, or if you are hard to wake up. |
Call your doctor at once if you have:
|
| Seek medical attention right away if you have symptoms of serotonin syndrome, such as: agitation, hallucinations, fever, sweating, shivering, fast heart rate, muscle stiffness, twitching, loss of coordination, nausea, vomiting, or diarrhea. |
| Serious side effects may be more likely in older adults and those who are overweight, malnourished, or debilitated. |
| Long-term use of opioid medication may affect fertility (ability to have children) in men or women. It is not known whether opioid effects on fertility are permanent. |
Common side effects may include:
|
| This is not a complete list of side effects and others may occur. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
Important Information:
Seizures have been reported in patients taking tramadol. Your risk of seizures is higher if you are taking higher doses of tramadol over what is recommended. Seizure risk is also higher in those with a seizure disorder or those taking certain antidepressants or opioid medications.
Tramadol should not be used if you are suicidal or prone to addiction.You should not take tramadol if you have severe breathing problems, a blockage in your stomach or intestines, or if you have recently used alcohol, sedatives, tranquilizers, narcotic medication, or an MAO inhibitor (isocarboxazid, linezolid, methylene blue injection, phenelzine, rasagiline, selegiline, tranylcypromine, and others).
Tramadol can slow or stop your breathing, and may be habit-forming. MISUSE OF THIS MEDICINE CAN CAUSE ADDICTION, OVERDOSE, OR DEATH, especially in a child or other person using the medicine without a prescription.
Tramadol should not be given to a child younger than 12 years old. Ultram ER should not be given to anyone younger than 18 years old.Taking tramadol during pregnancy may cause life-threatening withdrawal symptoms in the newborn.
Fatal side effects can occur if you use this medicine with alcohol, or with other drugs that cause drowsiness or slow your breathing.
Avoid drinking alcohol. Dangerous side effects could occur.
This medicine may impair your thinking or reactions. Avoid driving or operating machinery until you know how this medicine will affect you. Dizziness or severe drowsiness can cause falls or other accidents.
Interactions:
Drug Interactions (532) Alcohol/Food Interactions (1) Disease Interactions (10)
What other drugs will affect Tramadol?
You may have breathing problems or withdrawal symptoms if you start or stop taking certain other medicines. Tell your doctor if you also use an antibiotic, antifungal medication, heart or blood pressure medication, seizure medication, or medicine to treat HIV or hepatitis C. Tramadol can interact with many other drugs and cause dangerous side effects or death. Be sure your doctor knows if you also use:This list is not complete. Other drugs may interact with tramadol, including prescription and over-the-counter medicines, vitamins, and herbal products. Not all possible interactions are listed in this medication guide.
- cold or allergy medicines, bronchodilator asthma/COPD medication, or a diuretic ("water pill");
- medicines for motion sickness, irritable bowel syndrome, or overactive bladder;
- other narcotic medications - opioid pain medicine or prescription cough medicine;
- a sedative like Valium - diazepam, alprazolam, lorazepam, Xanax, Klonopin, Versed, and others;
- drugs that make you sleepy or slow your breathing - a sleeping pill, muscle relaxer, medicine to treat mood disorders or mental illness; or
- drugs that affect serotonin levels in your body - a stimulant, or medicine for depression, Parkinson's disease, migraine headaches, serious infections, or nausea and vomiting.
A total of 532 drugs are known to interact with Tramadol.
- 290 major drug interactions
- 239 moderate drug interactions
- 3 minor drug interactions
 Tramadol Interactions:
Tramadol Interactions:
https://www.drugs.com/tramadol.html
| Maximum Dosage: | |
|---|---|
| Prescribers Digital Reference | |

Ultram - Drug Summary: https://www.pdr.net/drug-summary/Ultram-tramadol-hydrochloride-950 | |
| Adults: | 400 mg/day PO for immediate-release and orally disintegrating tablets; 300 mg/day PO for extended-release formulations. |
| Geriatric: | 65 to 75 years: 400 mg/day PO for immediate-release and orally disintegrating tablets; 300 mg/day PO for extended-release formulations. Older than 75 years: 300 mg/day PO. |
| Adolescents: | 17 years: 400 mg/day PO for immediate-release and orally disintegrating tablets; 300 mg/day PO for dual-matrix extended-release tablets (Ryzolt). Safety and efficacy of other formulations have not been established. 16 years: 300 mg/day PO for dual-matrix extended-release tablets (Ryzolt). Safety and efficacy of other formulations have not been established. 13 to 16 years: Safety and efficacy have not been established. |
| Children: | 12 years: Safety and efficacy have not been established. 1 to 11 years: Use is contraindicated. |
| Infants: | Use is contraindicated. |
| Black Box Warnings: |
|---|

Ultram, ConZip (tramadol) (Rx): https://reference.medscape.com/drug/ultram-conzip-tramadol-343324 |
Opioid analgesic risk evaluation and mitigation strategy (REMS)
Addiction, abuse, and misuse
Life-threatening respiratory depression
Accidental ingestion
Neonatal opioid withdrawal syndrome
Ultra-rapid metabolism and other risk factors for life-threatening respiratory depression in children
Interactions with Drugs Affecting Cytochrome P450 Isoenzymes
Risks From Concomitant Use With Benzodiazepines Or Other CNS Depressants
|
Pediatric:
- Qdolo should not be used in children younger than 12 years of age and to relieve pain after surgery (eg, surgery to remove adenoid or tonsil) in children.
- Appropriate studies have not been performed on the relationship of age to the effects of Rybix ODT and Ryzolt in children younger than 16 years of age. Safety and efficacy have not been established.
- Conzip extended-release capsules and Ultram tablets should not be used in children younger than 12 years of age. It should not be used to relieve pain after surgery removal of tonsils or adenoids in any children.
Severe breathing problems and deaths have been reported in some children who received tramadol after tonsil or adenoid surgery.
- Appropriate studies have not been performed on the relationship of age to the effects of Ultram ER extended-release tablets in the pediatric population. Safety and efficacy have not been established.
Geriatric:
Appropriate studies performed to date have not demonstrated geriatric-specific problems that would limit the usefulness of tramadol in the elderly.However, elderly patients are more likely to have unwanted side effects (eg, constipation, lightheadedness, dizziness, or fainting, stomach upset, weakness) and age-related liver, kidney, heart, or lung problems, which may require caution and an adjustment in the dose for patients receiving tramadol.
 Tramadol (Oral Route) (ConZip | FusePaq Synapryn | Rybix ODT | Ryzolt | Ultram | Ultram ER):
Tramadol (Oral Route) (ConZip | FusePaq Synapryn | Rybix ODT | Ryzolt | Ultram | Ultram ER):
https://www.mayoclinic.org/drugs-supplements/tramadol-oral-route/description/drg-20068050
Breastfeeding:Summary of Use During Lactation:
The excretion of tramadol into milk is low and even lower amounts of the active metabolite, O-desmethyltramadol, are excreted. With usual maternal dosage, the amount excreted into breastmilk is much less than the dose that has been given to newborn infants for analgesia. A study of breastfeeding in breastfed newborn infants found no adverse effects attributable to tramadol. Although tramadol is unlikely to adversely affect nursing infant, the U.S. Food and Drug Administration and the manufacturer recommend against the use of tramadol during breastfeeding. If tramadol is used, monitor infants for increased sleepiness (more than usual), difficulty breastfeeding, breathing difficulties or limpness, and contact a physician immediately if any of these occur.Effects in Breastfed Infants:
Seventy-five breastfed infants whose mothers were breastfeeding and taking tramadol 100 mg every 6 hours following a cesarean section were compared to 75 matched infants at 2 to 4 days of age. Forty-nine percent of the mothers taking tramadol and all of the control mothers were taking other opiates (primarily oxycodone) and 61% of and 58%, respectively, also were taking a nonsteroidal antiinflammatory agent (primarily diclofenac). Examination by a pediatrician revealed no difference between the groups using the Neurologic and Adaptive Capacity Score.Effects on Lactation and Breastmilk:
Tramadol can increase serum prolactin. However, the prolactin level in a mother with established lactation may not affect her ability to breastfeed. A randomized study compared tramadol and naproxen for post-cesarean section pain. Patients received the drugs either on a fixed schedule or as needed. No difference in breastfeeding rates were seen among the groups. In a study in China, women with a scheduled cesarean section were randomized to receive intravenous patient-controlled analgesia with either sufentanil or tramadol. Postpartum prolactin levels were higher in the tramadol group (348 mcg/L) than in the sufentanil group (314 mcg/L). The onset of lactation was sooner in the tramadol group (21.4 hours) than in the sufentanil group (25.1 hours). Both of these difference were statistically significant.Injectable tramadol is not available in the United States.
Alternate Drugs to Consider:
- Acetaminophen
- Ibuprofen
- Morphine
 Tramadol Drug Levels and Effects:
Tramadol Drug Levels and Effects:
https://www.ncbi.nlm.nih.gov/books/NBK501260/

https://healthunlocked.com/tag/tramadol
What's the difference between Tramadol and Toradol? They're the drugs Bobby Portis says he mixed up, causing his suspension. - The agent for Bobby Portis is calling the Milwaukee Bucks forward's use of Tramadol, the drug that has led to his suspension, "an honest mistake." Portis has been suspended for the Bucks' next 25 ... Wednesday February 19, 2025 - sports.yahoo.com What is tramadol, how dangerous is it – and where is it illegal? - An Englishwoman named Laura Plummer is in jail in Egypt on suspicion of drug trafficking 290 tramadol tablets. The tablets (available on prescription in the UK) were found in her suitcase when it was ... Tramadol vs. Hydrocodone - Tramadol and hydrocodone are two types of potent pain relievers called opioid analgesics. Both work slightly differently and may cause different side effects. Tramadol is a synthetic opioid. Synthetic ... Killing Pain: Tramadol the 'Safe' Drug of Abuse - For years following spine surgery, Anthony Fort suffered. And like many with his condition, the 46-year-old gospel singer and former construction worker used a combination of medications to alleviate ... What is Tramadol, what are the side effects of the drug and is it illegal to buy in the UK? - TRAMADOL is the highly addictive painkiller which can only be prescribed by a doctor to patients suffering from severe pain. Here’s all you need to know about the drug Matt Cardle took at his lowest – ... Opioid use booming as tramadol crisis emerges in Africa - U.N. drug report - Add Yahoo as a preferred source to see more of our stories on Google. VIENNA, June 26 (Reuters) - Synthetic opioid use is booming, the United Nations said on Wednesday in a worldwide drug report that ... DEA: Pain Drug Tramadol Now a Controlled Substance - The Drug Enforcement Administration (DEA) has officially declared tramadol as a Class IV substance, including its salts, isomers, and salts of isomers. This new scheduling will go into effect August ... Tramadol: Rugby League's fight to kick 'dirty drug' - Tramadol was the "go-to" drug in rugby league, the BBC has been told Following an inquest into the death of young rugby league player Archie Bruce, the BBC has been told a powerful prescription-only ... What is Tramadol? What to know about the drug after Bobby Portis Jr.'s 25-game suspension - Milwaukee Bucks player Bobby Portis Jr. was suspended for 25 games for violating the NBA's anti-drug policy after testing positive for Tramadol. Portis and his agent claim the positive test was a ... Why football fears tramadol: ‘It’s an evil drug – it nearly killed me’ - Last week, every footballer in England and Wales received an email warning them about a painkiller that, until now, they might never have realised could put them at risk. The warning came from the ... What is Tramadol, what are the side effects of the drug and is it illegal to buy in the UK? - TRAMADOL is the highly addictive painkiller which can only be prescribed by a doctor to patients suffering from severe pain. Here’s all you need to know about the drug Matt Cardle took at his lowest – ...
| ||
| Opioids | Link to this page |





