[ Home ] [ Controlled Substances ] [ Depressants ]
SUVOREXANT
|
Until now, medications for insomnia have largely focused on enhancing sleep promoting mechanisms in the brain. However, the physiology of insomnia for most people is a problem of the alertness system not switching off. In the late 1990s orexin (also called hypocretin) was discovered as one of the key neurotransmitters involved in promoting wakefulness. Since then the pharmaceutical industry has been working on developing orexin antagonists (blockers) as treatment for insomnia. Suvorexant (brand name Belsomra) is the first of these to be approved for use.
Why is blocking orexin a potential insomnia treatment?
Orexin is considered a 'master controller' of the sleep wake control system. Orexin both stimulates the wake-promoting system, but also stabilises the switching between wake and sleep. As orexin levels rise in the morning people feel more alert. Then around the time of going to sleep at night, orexin levels fall resulting in people feeling less alert.
In the early 2000s, another important discovery highlighted the importance of orexin in maintaining stable wakefulness. People with the condition narcolepsy with cataplexy were found to have very low numbers of orexin neurones in the lateral hypothalamus and low or absent levels of orexin in the fluid surrounding the brain. This finding suggested that blocking orexin at night could induce sleepiness in people having trouble 'switching off', getting to sleep or waking through the night.
What should I expect if using suvorexant?
- Sleep may feel different: As the mechanism of suvorexant's action is different to the action of other sleep medications, you should expect that the sleep you experience taking suvorexant may be different from how sleep feels when taking other sleep medications.
- Sleep may not feel better immediately: If switching from another type of sleeping tablet to suvorexant, there may be a period of rebound insomnia, or worsening insomnia symptoms, from stopping the other medication. In addition, in the trials of suvorexant, it took some time for people's experience of sleep to improve even though sleep measured on a sleep study improved on the first night after taking suvorexant.
- Effects may occur at different parts of the night: Whilst suvorexant does help people get to sleep faster, it's main effects appear to be throughout the night, even in the second half of the night when other medications often have less of an effect. Suvorexant may not cause people to wake less, but seems to help people get back to sleep more quickly increasing the overall amount of sleep.

https://sleephub.com.au/suvorexant/
Why is it prescribed:
Suvorexant is used treat insomnia (difficulty falling asleep or staying asleep). Suvorexant is in a class of medications called orexin receptor antagonists. It works by blocking the action of a certain natural substance in the brain that causes wakefulness.
Suvorexant
2013 Article in The New Yorker:
Suvorexant, which Merck describes as "rationally designed" - rather than stumbled upon, like most drugs - influences a more precise set of neurotransmitters and receptors. Orexin neurotransmitters, first identified fifteen years ago, promote wakefulness. When suvorexant is in the brain, orexin is less likely to reach orexin receptors. Instead of promoting general, stupefying brain inactivity, suvorexant aims at standing in the way of a keep-awake signal. This difference may or may not come to mean a lot to insomniacs, but Merck's marketing is likely to encourage the perception that suvorexant ends the dance by turning off the music, whereas a drug like Ambien knocks the dancer senseless.

https://www.newyorker.com/magazine/2013/12/09/the-big-sleep-2
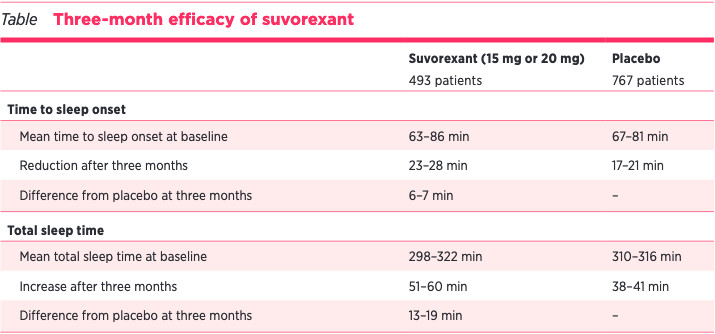
There are different patterns of insomnia, such as a delayed onset of sleep and difficulty maintaining sleep. Orexins are neuropeptides which are involved in the regulation of sleep and arousal. Orexins A and B promote wakefulness. Blocking their receptors should therefore reduce wakefulness and promote sleep. The drug is taken within 30 minutes of bedtime. This should be at least seven hours before the patient plans to get up again. The maximum drug concentration is reached in two hours. Suvorexant is metabolised with most of the metabolites being excreted in the faeces. Its half-life is approximately 12 hours.

https://www.nps.org.au/australian-prescriber/articles/suvorexant-for-insomnia


https://www.nps.org.au/assets/0db6520f14f6939e-94e8bb8573f4-suvorexant-for-insomnia_40-116.pdf
Dosage Forms & Strengths:
Tablet: Schedule IV:
- 5mg
- 10mg
- 15mg
- 20mg
Insomnia:
Indicated for insomnia characterized by difficulties with sleep onset and/or sleep maintenance. Recommended starting dose: 10 mg PO taken no more than once per night and within 30 minutes of going to bed, with at least 7 hr remaining before the planned time of awakening. Use the lowest dose effective for the patient If 10 mg dose is well-tolerated but not effective, the dose can be increased. Not to exceed 20 mg once daily

https://reference.medscape.com/drug/belsomra-suvorexant-999859
American Family Physician 2016:
Unlike other hypnotics, it blocks the binding of neuropeptides (orexin A and B) that promote wakefulness.
Safety:
Suvorexant carries a slight risk of abuse. Events that suggested possible abuse were identified in 3% to 4% of patients in clinical trials. Suvorexant is a central nervous system depressant. Rare side effects include sleep paralysis, hypnagogic hallucinations, and mild cataplexy, consisting of brief periods of leg weakness. Suvorexant should be avoided in patients with depression because it may worsen symptoms; patients with severe depression were excluded from clinical trials. It should also not be used in patients with narcolepsy, obstructive sleep apnea, or severe chronic obstructive pulmonary disease. It is a U.S. Food and Drug Administration pregnancy category C drug. It is unknown whether suvorexant is excreted in human breast milk.
Tolerability:
Suvorexant causes significant somnolence in 7% to 11% of patients, and driving performance is impaired in up to 20% of patients, especially at higher doses and when starting the medication. Performance can be affected without the patient being aware.
Effectiveness:
Suvorexant has been compared with placebo for treatment of chronic insomnia in patients older than 50 years who slept less than 5.5 hours per night on average. Using objective measures, consistent use for one month will decrease the time to fall asleep from an average of 65 to 69 minutes to 32 to 35 minutes, although this change is only eight to 10 minutes less than when using placebo. It will also help patients stay asleep to a greater extent than placebo.
Price:
Suvorexant costs approximately $305 for a one-month supply of 5-, 10-, 15-, or 20-mg tablets.
Simplicity:
The recommended starting dosage of suvorexant is 10 mg once daily. It should be taken within 30 minutes of going to bed with at least seven hours remaining before the planned time of awakening.
Bottom Line:
Suvorexant effectively increases total sleep time and decreases time to sleep onset. Because of its risk of abuse, significant incidence of next-day impairment, and high cost, it should be reserved for use in patients for whom other insomnia therapies, such as cognitive behavior therapy and lifestyle changes, have failed.

https://www.aafp.org/afp/2016/0615/p1016.html


https://www.aafp.org/afp/2016/0615/afp20160615p1016.pdf
Next-day impairment:
The drug is available in 5-, 10-, 15-, and 20-mg strengths. Recommended dose is 10 mg. Total dose should not exceed 20 mg once daily because suvorexant, like other sleep medications, can impair daytime wakefulness and driving skills and may increase the risk of falling asleep while driving. The risk is greater if suvorexant is taken with less than a full night of sleep remaining, at higher than the recommended dose, or if co-administered with other central nervous system depressants or other drugs that increase blood levels of suvorexant.
Patients taking suvorexant may engage in complex behaviors while not being fully awake, such as preparing and eating food, making phone calls, or having sex. Chances of such activity increase if patients have consumed alcohol or taken other medicines that make them sleepy.

https://www.pharmacytoday.org/article/S1042-0991(15)30656-3/fulltext
Alzheimer Disease:
Feb 4, 2020 - The FDA has approved an expanded indication for suvorexant (Belsomra; Merck) for the treatment of insomnia in patients with mild to moderate Alzheimer disease (AD).
The update stems from results from a randomized, double-blind, placebo-controlled, parallel-group, 4-week polysomnography trial that evaluated the safety and efficacy of suvorexant in patients with mild to moderate AD.2 A total of 285 patients were randomly assigned 1:1 to either 10 mg of suvorexant or placebo. Among the 142 patients who received suvorexant, 77% received an increased dose of 20 mg suvorexant for approximately 14 additional days. Compared to those treated with placebo, patients taking suvorexant demonstrated a statically significant improvement in both total sleep time (TST) and wake after sleep onset outcomes.
https://www.neurologylive.com/view/suvorexant-now-indicated-for-treatment-of-insomnia-in-alzheimer-disease
Alzheimer Disease:
Many people with AD suffer from disruption of circadian rhythms that leads to poor sleep, nighttime activity and daytime sleepiness. Circadian disruption is a frequent cause of institutionalization. Changes in sleep/wake cycles also occur in preclinical AD, and are linked to increased amyloid deposition and risk of cognitive decline. Thus, sleep aids are of interest as potential disease modifiers. CSF orexin levels are elevated in Alzheimer's disease. In the APP/PS1 mouse model of AD, suvorexant was shown to improve circadian rhythms and cognitive function, restore hippocampal synaptic plasticity, and reduce AB plaque deposition in hippocampus and cortex.

https://www.alzforum.org/therapeutics/suvorexant
Orexin Receptors:
Belsomra has a unique mechanism of action different from other sleep medications, for example, from benzodiazepine type of sleep medicines like diazepam (Valium).
Belsomra causes sleep by blocking receptors in the brain called orexin receptors. Orexin A and B are brain chemicals that cause people to stay awake by binding to orexin receptors. Belsomra prevents orexin A and orexin B from causing wakefulness by blocking their attachment to orexin receptors. Reduced wakefulness means more sleep or increased ability to fall asleep.

https://www.medicinenet.com/suvorexant_belsomra/article.htm
Suvorexant is a selective dual antagonist of orexin receptors OX1R and OX2R. It has been approved for the treatment of insomnia. The mechanism by which suvorexant exerts its therapeutic effect in insomnia is presumed to be through antagonism of orexin receptors. The orexin neuropeptide signaling system is a central promoter of wakefulness. Blocking the binding of wake-promoting neuropeptides orexin A and orexin B to receptors OX1R and OX2R is thought to suppress wake drive.
 Suvorexant:
Suvorexant:
https://drugs.ncats.io/drug/081L192FO9
| Maximum Dosage: | |
|---|---|
| Prescribers Digital Reference | |

Belsomra - Drug Summary: https://www.pdr.net/drug-summary/Belsomra-suvorexant-3605 | |
| Adults: | 20 mg/day PO. |
| Geriatric: | 20 mg/day PO. |
| Adolescents: | Safety and efficacy have not been established. |
| Children: | Safety and efficacy have not been established. |
| Infants: | Not indicated. |
| Neonates: | Not indicated. |
Prescribed for:
- Difficulty sleeping
Used for:
This medication is used to treat a certain sleep problem (insomnia). It may help you fall asleep and stay asleep longer, so you can get a better night's rest. Suvorexant belongs to a class of drugs known as sedative-hypnotics.If your insomnia continues for longer than 7 to 10 days after starting treatment, talk to your doctor to see if you need other treatment.
Before using:
Tell your doctor or pharmacist your medical history, especially of:
- a certain sleep disorder (narcolepsy)
- liver disease
- lung disease/breathing problems (such as sleep apnea, chronic obstructive pulmonary disease- COPD)
- mental/mood problems (such as depression, thoughts of suicide)
- personal or family history of a substance use disorder (such as overuse of or addiction to drugs/alcohol)
- a certain muscle weakness disorder (cataplexy)
Precautions:
- The effects of this drug can last even after you wake up the next day.
- If you did not get at least 7 hours of sleep or took other medications that made you sleepy or are more sensitive to this drug, you may feel alert but not think clearly enough to drive.
- Alcohol or marijuana (cannabis) can also make you more sleepy.
- Wait at least 8 hours after taking this drug before driving, and do not drive, use machinery, or do anything that needs alertness until you can do it safely.
- Avoid alcoholic beverages.
- Talk to your doctor if you are using marijuana (cannabis).
- If you take the 20 milligram dosage of suvorexant, do not drive, use machinery, or do anything that needs alertness the next day.
 Suvorexant Tablet (Belsomra):
Suvorexant Tablet (Belsomra):
https://www.webmd.com/drugs/2/drug-166796/suvorexant-oral/details
What is the most important information I should know about suvorexant?
- You should not use suvorexant if you have narcolepsy.
| Side Effects: |
|---|
| |
Belsomra (suvorexant tablets): https://www.rxlist.com/belsomra-drug.htm |
Common side effects of Belsomra include:
|
| Belsomra may cause serious side effects that you may not know are happening to you, including "sleep-walking" or doing other activities when you are asleep like eating, talking, having sex, or driving a car. Call your doctor right away if you find out you have done any of these activities after taking Belsomra. |
Pediatric:
Appropriate studies have not been performed on the relationship of age to the effects of suvorexant in the pediatric population.Safety and efficacy have not been established.
Geriatric:
Appropriate studies performed to date have not demonstrated geriatric-specific problems that would limit the usefulness of suvorexant in the elderly.Other Interactions:
- Grapefruit Juice
Other Medical Problems:
The presence of other medical problems may affect the use of this medicine. Make sure you tell your doctor if you have any other medical problems, especially:
- Alcohol abuse, history of
- Drug abuse, history of - Dependence on suvorexant may be more likely to develop
- Breathing or lung problems (eg, COPD, sleep apnea)
- Cataplexy (sudden onset of muscle weakness), history of
- Depression, history of
- Mental illness, history of - Use with caution. May make these conditions worse
- Narcolepsy (uncontrollable desire for sleep or a sudden attack of deep sleep) - Should not be used in patients with this condition
- Obese patients (especially in obese women) - Use with caution. The effects may be increased because of slower removal of the medicine from the body
 Suvorexant (Oral Route) (Belsomra):
Suvorexant (Oral Route) (Belsomra):
https://www.mayoclinic.org/drugs-supplements/suvorexant-oral-route/description/drg-20113017
Important Information
Do not take more suvorexant than your doctor has prescribed.
Take suvorexant 30 minutes before bedtime. Never take suvorexant if you do not have 7 hours to sleep before being active again.Some people using this medicine have engaged in activity while not fully awake and later had no memory of it. If this happens to you, call your doctor right away.
For at least 8 hours after taking suvorexant, do not drive or do anything that requires you to be alert. Your reactions may be impaired.
Avoid drinking alcohol. Do not take suvorexant if you have consumed alcohol within a few hours before getting ready for sleep.
Interactions:
Drug Interactions (418) Alcohol/Food Interactions (1) Disease Interactions (6)
What other drugs will affect Suvorexant?
Using suvorexant with other drugs that make you drowsy can worsen this effect. Ask your doctor before using opioid medication, a sleeping pill, a muscle relaxer, or medicine for anxiety or seizures. Sometimes it is not safe to use certain medications at the same time. Some drugs can affect your blood levels of other drugs you take, which may increase side effects or make the medications less effective. Other drugs may affect suvorexant, including prescription and over-the-counter medicines, vitamins, and herbal products. Tell your doctor about all your current medicines and any medicine you start or stop using.A total of 418 drugs are known to interact with Suvorexant.
- 77 major drug interactions
- 331 moderate drug interactions
- 10 minor drug interactions
 Suvorexant Information:
Suvorexant Information:
https://www.drugs.com/mtm/suvorexant.html
Liver:
Suvorexant therapy is associated with rare occurrence of transient serum enzyme elevations, but has not been implicated in cases of clinically apparent liver injury.
Suvorexant Hepatotoxicity:
In several clinical trials, suvorexant was found to be well tolerated, with serum ALT elevations in 0 to 5% of patients, usually with higher doses, and resolving spontaneously without dose modification. In the registration trials of suvorexant, there were no reports of clinically apparent liver injury. Suvorexant has been available for a limited period of time, but has yet to be implicated in causing clinically apparent liver injury.E Likelihood score: E (unlikely cause of clinically apparent liver injury).
 Suvorexant Overview:
Suvorexant Overview:
https://www.ncbi.nlm.nih.gov/books/NBK547900/
Consumer Reports:
Here's why you can skip the new insomnia drug, Belsomra
It's expensive, barely helps, and poses safety concerns:A sleepless night or two can leave you so tired and miserable that it can be tempting to take a medication that promises to help you slip into slumber. But the truth is that sleeping pills like Ambien and Lunesta don't actually improve your sleep much, according to a Consumer Reports Best Buy Drugs analysis, and the newest insomnia medication, Belsomra (suvorexant), is no exception. It might help you nod off a few minutes faster or stay asleep slightly longer. But that small benefit comes with some big safety concerns, such as being too drowsy to drive the next day or feeling like you can't move or talk.
We were prompted to take a close look at Belsomra, which is made by Merck, because it's a new type of sleeping pill called an orexin-receptor antagonist. It acts on the brain in a different way compared to older insomnia meds. The Food and Drug Administration initially rejected high doses of Belsomra - 30 mg and 40 mg - because it said they posed a dangerous risk of next-day drowsiness that could lead to deadly auto crashes. The FDA eventually approved lower doses of the drug - 5 mg, 10 mg, and 20 mg.
People who took a 15 mg or 20 mg dose of Belsomra every night for 3 months fell asleep just 6 minutes faster on average than those who got a placebo pill. And the Belsomra group slept only 16 minutes longer - 6 hours and 12 minutes total vs. 5 hours and 56 minutes for the placebo group. Those small improvements in sleep didn't translate to people feeling more refreshed. Instead, more people who took Belsomra felt drowsy the next day compared with those who took a placebo.
Bottom Line:
Our medical advisers say that a sleeping pill is usually not the best treatment for insomnia. Instead, cognitive behavioral therapy, which involves talking to a therapist to learn a new set of behaviors regarding sleep, is as effective as sleeping pills, and has been shown to help up to 80 percent of chronic insomnia sufferers get some shuteye. Studies have also found that improving your sleep habits, such as relaxation training, setting and sticking to consistent bedtimes and wake-up times, regular exercise, quitting smoking, cutting back on caffeine and alcohol in the afternoon and evening, keeping your bedroom quiet and dark, and not watching TV or using computers in bed can help relieve insomnia.
 Here's why you can skip the new insomnia drug, Belsomra (2015):
Here's why you can skip the new insomnia drug, Belsomra (2015):
https://www.consumerreports.org/cro/news/2015/07/skip-new-insomnia-drug-belsomra/index.htm
Complaints About Belsomra:
 FDA Fields Complaints on Sleeping Pill Suvorexant (2016):
FDA Fields Complaints on Sleeping Pill Suvorexant (2016):
https://www.consumerreports.org/drugs/fda-fields-complaints-on-sleeping-pill-suvorexant-belsomra/
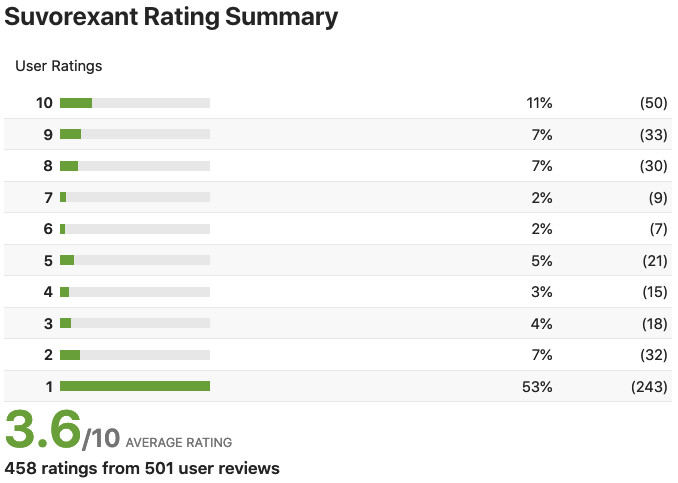
 User Reviews for Suvorexant to treat Insomnia:
User Reviews for Suvorexant to treat Insomnia:
https://www.drugs.com/comments/suvorexant/for-insomnia.html

https://www.everydayhealth.com/drugs/belsomra/reviews

https://www.reddit.com/search/?q=Suvorexant
Belsomra Reddit Search:
https://www.reddit.com/search/?q=Belsomra

https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=e5b72731-1acb-45b7-9c13-290ad12d3951


http://www.alzdiscovery.org/uploads/cognitive_vitality_media/Suvorexant-Cognitive-Vitality-For-Researchers.pdf


https://www.merck.com/product/usa/pi_circulars/b/belsomra/belsomra_mg.pdf


https://www.merck.com/product/usa/pi_circulars/b/belsomra/belsomra_pi.pdf


https://www.accessdata.fda.gov/drugsatfda_docs/label/2014/204569s000lbledt.pdf


https://www.pbm.va.gov/PBM/clinicalguidance/drugmonographs/Suvorexant_monograph_FINAL.pdf
- Medication for the treatment of insomnia characterized by difficulties with sleep onset and/or sleep maintenance
- Approved for sale by the U.S. Food and Drug Administration (FDA) in 2014
- The U.S. Drug Enforcement Administration (DEA) placed it on the list of schedule IV controlled substances because it may lead to limited physical dependence or psychological dependence. It produces similar reinforcing effects to those of zolpidem and thus may have a similar abuse liability. As such, suvorexant has been designated a schedule IV.
- The potential for psychological dependence is similar to that of zolpidem as well
It is unclear how the medication compares to others used for insomnia as no comparisons have been done. It is also unclear if this medication is safe among people with a history of addiction, as they were excluded from the clinical trials of suvorexant.
- Suvorexant is not recommended in people with liver impairment
- May cause fetal harm during pregnancy
- Suvorexant is contraindicated in people diagnosed with narcolepsy
Side Effects:
The most common complaint about the drug is from users who report that it did not help them to sleep. Some people reported that the drug caused a sleep disturbance such as a nightmare, sleep terror, or abnormal dream. Others reported that the drug caused them to be more awake. Issues include sleepiness the next day and issues with driving. Other concerns include thoughts of suicide- The elimination half-life is reported to be 12 hours
Initially released November 2014 in Japan, then later reached the United States in February 2015, Australia in November 2016, and Canada in November 2018
Suvorexant - Indications, Dosage, Side Effects and Precautions - Discover comprehensive details about Suvorexant, including its pronunciation, uses, dosage instructions, indications, and guidelines on how and when to take it or avoid it. The updated prescription ... Thursday November 07, 2024 - medindia.net FDA Fields Complaints on Sleeping Pill Suvorexant - When you shop through retailer links on our site, we may earn affiliate commissions. 100% of the fees we collect are used to support our nonprofit mission. Learn more. One way to do that is through ... How Does Insomnia Drug Suvorexant Block Oxycodone Relapse? - ‘In prescription opioid abuse, repurposing insomnia medication suvorexant is an effective approach for decreasing drug intake and blocking relapse. ’ How suvorexant has a beneficial effect on OUD is ... FDA: Merck's sleep drug suvorexant effective, but safety issues draw heavy flak - FDA staffers found clear evidence to prove that Merck's blockbuster drug candidate suvorexant is quite effective in promoting sleep, but the internal review cites multiple safety issues for the high ... Suvorexant may help those with Alzheimer’s ease insomnia - The drug suvorexant, sold as Belsomra, improves total sleep time in adults with probable Alzheimer’s disease and insomnia, according to a recent study published online in Alzheimer’s & Dementia. Sleep ... FDA raises concerns about experimental sleep aid suvorexant - WASHINGTON While an experimental insomnia drug from Merck was effective at helping patients sleep, it also had dangerous side effects including daytime drowsiness and suicidal thinking, a Food and ... As FDA Decision Loomed For Suvorexant, Chemists Discussed Sleep Drug Field’s Big Questions - Coleman, an executive director of discovery chemistry at Merck, returned to the City by the Bay three weeks ago to lead a symposium about that very mechanism. His session in the Division of Medicinal ... FDA OKs New Kind of Sleep Drug Suvorexant (Belsomra) - The US Food and Drug Administration (FDA) today approved a first-in-class insomnia drug called suvorexant (Belsomra, Merck) after the manufacturer lowered the dosages to satisfy the agency's safety ... Merck Announces FDA Acceptance of New Drug Application for Suvorexant, an Investigational Insomnia Medicine - WHITEHOUSE STATION, N.J.--(BUSINESS WIRE)--Merck (NYSE: MRK), known as MSD outside the United States and Canada, today announced that the New Drug Application (NDA) for suvorexant, the company's ... Effects of Suvorexant, a Dual Orexin/Hypocretin Receptor Antagonist, on Impulsive Behavior Associated with Cocaine - Male Sprague-Dawley rats (Charles River) arrived at Temple University’s vivarium ~60 days old, were pair-housed and food-restricted to 85% of their free-fed bodyweight. Rats were acclimated for at ... FDA Fields Complaints on Sleeping Pill Suvorexant - A recent analysis of more than 1,000 safety reports submitted to the Food and Drug Administration about the sleeping pill suvorexant (Belsomra), found that it was associated with side effects such as ... Merck Announces FDA Acceptance of New Drug Application for Suvorexant, an Investigational Insomnia M - WHITEHOUSE STATION, N.J.--(BUSINESS WIRE)-- Merck (NYS: MRK) , known as MSD outside the United States and Canada, today announced that the New Drug Application (NDA) for suvorexant, the company's ...
| ||
| Depressants | Link to this page |









