[ Home ] [ Controlled Substances ] [ Prescription ]
CARISOPRODOL
|
A centrally acting skeletal muscle relaxant whose mechanism of action is not completely understood but may be related to its sedative actions. Carisoprodol is indicated for the relief of discomfort associated with acute, painful musculoskeletal conditions in adults. Most common adverse reactions (incidence > 2%) are drowsiness, dizziness, and headache. Carisoprodol might be mixtured with Aspirin and Codeine Phosphate. Studies indicating increased risk of abuse or addiction led to withdrawal of the drug from the market in Norway and other EU countries in 2008.
 Carisoprodol:
Carisoprodol:
https://drugs.ncats.io/drug/21925K482H
Carisoprodol is used to treat muscle pain. Carisoprodol is a prescription drug. It comes in the form of a tablet you take by mouth. Carisoprodol is available as a brand-name drug called Soma. It's also available as a generic drug. Carisoprodol only comes in the form of a tablet you take by mouth. Carisoprodol can cause drowsiness. The use of carisoprodol can cause dependence (addiction). Carisoprodol may cause seizures. This risk is higher if it's used with several drugs that are misused or used in overly high doses. Carisoprodol is used as a short-term treatment for muscle pain. This drug should only be taken for up to two to three weeks. Carisoprodol can make you drowsy. The use of drinks that contain alcohol can make your drowsiness worse. If you drink alcohol, talk to your doctor about whether this drug is safe for you.

https://www.healthline.com/health/carisoprodol-oral-tablet
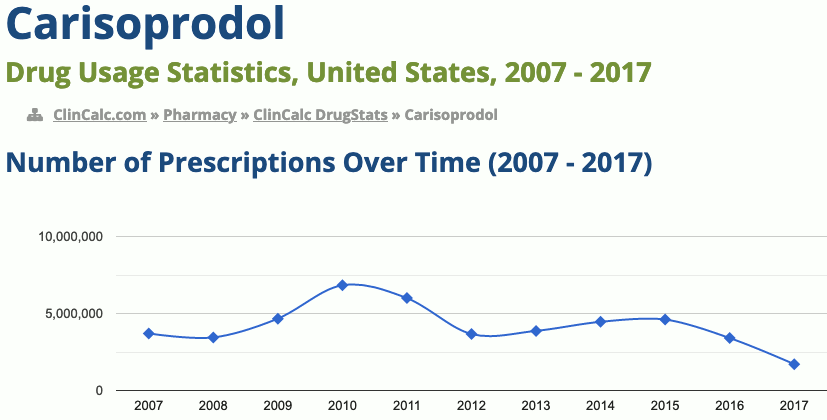
255th most prescribed medicine in the United States for 2017

https://clincalc.com/DrugStats/Drugs/Carisoprodol
Marketed since 1959

https://www.everydayhealth.com/drugs/carisoprodol/reviews
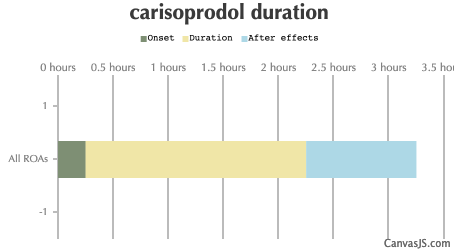
Use is only approved for up to three weeks. Effects generally begin within half an hour and last for up to six hours. Its approval in Europe was withdrawn in 2008. In the United States the wholesale costs is less than 0.10 USD per dose. In 2016 it was the 181st most prescribed medication in the United States with more than 3 million prescriptions.
Carisoprodol produces the same enzyme-inducing effects as some barbiturates and may be used to potentiate pro-drug opioids (Glutethimide and codeine), most commonly codeine and hydrocodone, by increasing the amount of pro-drug converted into the active metabolites (morphine and hydromorphone, respectively, although only a very small amount of hydrocodone is metabolized into hydromorphone via hepatic oxidation, whereas a large amount of codeine is metabolized into morphine via hepatic enzymes; codeine would have little analgesic effect without its metabolism into its far more potent relative, morphine).
This mechanism, in conjunction with the combined euphoria, sedation, and anxiolytic effects of the two drugs makes the practice incredibly dangerous and unintentional overdoses are common. The combination of hydrocodone and carisoprodol is referred to in some harm-reduction forums as a "Las Vegas cocktail". Many overdoses have resulted from recreational users combining these drugs to combine their individual effects without being aware of the enzyme-induction induced potentiation.


https://www.accessdata.fda.gov/drugsatfda_docs/label/2009/011792s043lbl.pdf
| Side Effects: |
| Get emergency medical help if you have signs of an allergic reaction: hives; difficult breathing; swelling of your face, lips, tongue, or throat. |
|---|
| RxList |
Soma (carisoprodol): https://www.rxlist.com/soma-drug/patient-images-side-effects.htm |
Stop using carisoprodol and call your doctor at once if you have:
|
Common side effects may include:
|
| This is not a complete list of side effects and others may occur. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
Pediatric:
Appropriate studies have not been performed on the relationship of age to the effects of carisoprodol in children below 16 years of age.Safety and efficacy have not been established.
Geriatric:
Appropriate studies on the relationship of age to the effects of carisoprodol have not been performed in the geriatric population.Safety and efficacy have not been established.
Other Medical Problems:
Make sure you tell your doctor if you have any other medical problems, especially:
- Drug abuse or dependence, or history of - Use with caution. Dependence on carisoprodol may develop
- Kidney disease
- Liver disease - Use with caution. The effects may be increased because of slower removal of the medicine from the body
- Porphyria (an enzyme problem), history of - Should not be used in patients with this condition
- Seizures - Use with caution. May make this condition worse
 Carisoprodol (Oral Route) (Soma - Vanadom):
Carisoprodol (Oral Route) (Soma - Vanadom):
https://www.mayoclinic.org/drugs-supplements/carisoprodol-oral-route/description/drg-20071941
Breastfeeding:Summary of Use During Lactation:
If carisoprodol is required by the mother, it is not necessarily a reason to discontinue breastfeeding.
Slight sedation has occurred in a breastfed newborn infant who was exposed during pregnancy and lactation; sedation might be more pronounced in newborns who are exposed for the first time during nursing.Other agents may be preferred, especially while nursing a newborn or preterm infant, or when other drugs that can cause sedation are used simultaneously.
Drug Levels:
Carisoprodol is metabolized almost entirely to meprobamate which contributes to the overall sedative effect.Carisoprodol has a half-life of 4 hours and meprobamate has a half-life of about 10 hours.
 Carisoprodol Drug Levels and Effects:
Carisoprodol Drug Levels and Effects:
https://www.ncbi.nlm.nih.gov/books/NBK501205/
| Duration: A skeletal muscle relaxant drug also known as Soma, carisoprodol has limited recreational value, however its main metabolite meprobamate has some moderate tranquilising properties. Sometimes found in concoctions with codeine or caffeine. NOTE: Tolerance raises very fast | |||
| Route | Onset | Duration | After Effects |
|---|---|---|---|
| Tripsit Factsheets | |||

Carisoprodol Basic Information: http://drugs.tripsit.me/carisoprodol | |||
| All ROAs: | 15-30 minutes | 2-4 hours | 1-3 hours |
| |||
Aliases:
| |||
| Effects: Anxiolytic, Sedative, Muscle Relaxant, Amnesia, Dystaxia, Hypnotic. | |||
| Maximum Dosage: | |
|---|---|
| Prescribers Digital Reference | |

Soma (carisoprodol) - Drug Summary: https://www.pdr.net/drug-summary/Soma-carisoprodol-2128 | |
| Adults: | 1,400 mg/day PO. |
| Geriatric: | 65 years: 1,400 mg/day PO. |
| Geriatric: | 66 years and older: Not recommended. |
| Adolescents: | 16 years and older: 1,400 mg/day PO. |
| Adolescents: | Less than 16 years: Safety and efficacy have not been established. |
| Children: | Safety and efficacy have not been established. |
| Infants: | Safety and efficacy have not been established. |
Liver:
Carisoprodol is a centrally acting muscle relaxant that has been in use for more than fifty years without significant evidence for causing hepatic injury.
Carisoprodol Hepatotoxicity:
There have been no adequate prospective studies demonstrating the rates of aminotransferase elevations on carisoprodol therapy or convincing case reports of clinically apparent liver injury due to carisoprodol. Thus, the hepatotoxic potential of this medication is low. It has been increasingly reported as a substance of abuse, taken in higher than recommended doses.E Likelihood score: E (unlikely cause of clinically apparent liver injury).
 Carisoprodol Overview:
Carisoprodol Overview:
https://www.ncbi.nlm.nih.gov/books/NBK548829/
Important Information:
You should not take carisoprodol if you have porphyria (a genetic enzyme disorder that causes symptoms affecting the skin or nervous system).Carisoprodol may be habit-forming. Never share this medicine with another person.
Misuse of habit-forming medicine can cause addiction, overdose, or death.
Carisoprodol can cause side effects that may impair your thinking or reactions. Be careful if you drive or do anything that requires you to be awake and alert. Avoid drinking alcohol. It can increase drowsiness and dizziness caused by this medicine.You may have withdrawal symptoms when you stop using this medicine after using it over a long period of time. Do not stop using this medication suddenly without first talking to your doctor. You may need to use less and less before you stop the medication completely.
Do not drink alcohol. Dangerous side effects or death could occur.
Avoid driving or hazardous activity until you know how this medicine will affect you. Dizziness or drowsiness can cause falls, accidents, or severe injuries.
Interactions:
Drug Interactions (330) Alcohol/Food Interactions (1) Disease Interactions (3)
What other drugs will affect Carisoprodol?
Sometimes it is not safe to use certain medications at the same time. Some drugs can affect your blood levels of other drugs you take, which may increase side effects or make the medications less effective.Using carisoprodol with other drugs that make you sleepy or slow your breathing can cause dangerous side effects or death. Ask your doctor before using opioid medication, a sleeping pill, a muscle relaxer, or medicine for anxiety or seizures.
Many drugs can interact with carisoprodol. This includes prescription and over-the-counter medicines, vitamins, and herbal products. Not all possible interactions are listed here. Tell your doctor about all your current medicines and any medicine you start or stop using.A total of 330 drugs are known to interact with Carisoprodol.
- 24 major drug interactions
- 305 moderate drug interactions
- 1 minor drug interaction
 Carisoprodol Drug Information:
Carisoprodol Drug Information:
https://www.drugs.com/carisoprodol.html


https://www.justice.gov/archive/ndic/pubs10/10913/10913p.pdf
Carisoprodol:
History:
On 1 June 1959, several American pharmacologists convened at Wayne State University in Detroit, Michigan to discuss a new drug. The drug, originally thought to have antiseptic properties, was found to have central muscle-relaxing properties. It had been developed by Frank Berger at Wallace Laboratories and was named carisoprodol. Carisoprodol was a modification of meprobamate, intended to have better muscle relaxing properties, less potential for abuse, and less risk of overdose. The substitution of one hydrogen atom with an isopropyl group on one of the carbamyl nitrogens was intended to yield a molecule with new pharmacological properties.
- A medication used to relax muscles after strains, sprains and muscle injuries.
- How it works is not clear. Some of its effects are believed to occur following being converted into meprobamate
- Approved for medical use in the United States in 1959
- Its approval in Europe was withdrawn in 2008
- It is taken orally
- Use is only approved for up to three weeks
- Effects generally begin within half an hour and last for up to six hours
- Common side effects include headache, dizziness, and sleepiness
- Serious side effect may include addiction, allergic reactions, and seizures
- It is available as a generic medication
- In 2017, it was the 255th most commonly prescribed medication in the United States, with more than one million prescriptions
- Recreational users of carisoprodol usually seek its potentially heavy sedating, relaxant, and anxiolytic effects.
- Because of its potentiating effects on narcotics, it is often abused in conjunction with many opioid drugs.
- It is not detected on standard drug testing screens.
Combining a muscle relaxant like carisoprodol with opioids and benzodiazepines is referred to as "The Holy Trinity" as it has been reported to increase the power of the "high".
Carisoprodol/aspirin/codeine - Carisoprodol 200mg, aspirin 325mg, codeine phosphate 16mg; tabs; contains sulfites. GI complication (eg, bleeding, perforation, obstruction) due to aspirin use ... Friday October 15, 2021 - empr.com Carisoma (Carisoprodol) Drug Price and Information - The Price section compares costs of the same generic drugs across brands and is purely for information purpose. Medindia neither buys nor sells drugs. Carisoprodol (Carisoma) is a muscle relaxant, for ... Withdrawal of export NOCs, manufacturing licences for tapentadol-carisoprodol combinations ordered - In a communication, the DCGI has asked the drug control authorities of the states and Union territories to immediately withdraw all export NOCs and manufacturing licences granted for any combination ... Govt bans production, export of drugs containing tapentadol and carisoprodol - Concerned over the potential for severe harm, the DCGI has called for the immediate withdrawal of all export NOCs and manufacturing permissions for tapentadol and carisoprodol combinations.(Reuters) ... Carisol (Carisoprodol) Drug Price and Information - Carisoprodol (Carisol) is a muscle relaxant, for the relief of discomfort associated with acute, painful musculoskeletal conditions. It blocks pain sensations between the nerves and the brain. The ...
| ||
| Prescription | Link to this page |






