[ Home ] [ Controlled Substances ] [ Hallucinogens ]
TMA
|
There are six possible analogues of TMA:
3,4,5 trimethoxyamphetamine
Dose: 100-250 mg Duration: 6-8 h
Effects: Similar to mescaline but perhaps less pleasant. More active.
This is the amphetamine analogue of mescaline, and may be derrived from elemicin, a fraction of nutmeg oil.
2,4,5 trimethoxyamphetamine
Dose: 20-40 mg Duration: 8-12 h
Effects: Strongly psychedelic, somewhat similar to mescaline. Pleasant.
Derrived from assarone, a component of calamus oil.
2,3,4 trimethoxyamphetamine
Not found to be active at 100 mg
2,3,5 trimethoxyamphetamine
Slight activity (~= 50 ug LSD) found at 80 mg ca. 6 h
2,3,6 trimethoxyamphetamine
Mild stimulant and psychedelic effects found at 30 mg (~75 ug LSD) 8-10 h
2,4,6 trimethoxyamphetamine
Dose: 25-50 mg Duration: 12-16 h
Effect: Very pleasant stimulant and psychedelic
 TMA-2 (and other TMAs) Information:
TMA-2 (and other TMAs) Information:
https://www.erowid.org/chemicals/tma2/tma2.shtml
TMA-1 was the first psychotomimetic drug synthesized in 1947 by modifying the structure of mescaline on the basis of the principles discovered in studying the relationships between chemical structures and biological activity, and has about twice the potency of mescaline.
TMA-2, the geometric isomer of TMA-1, was first synthesized in 1933, and its psychotomimetic properties were first reported by Shulgin in 1964. This was thus called TMA-2 as the second of the six possible positional isomers found to be psychotomimetic. TMA-2 was reported to possess some 20 times the potency of mescaline and is the most active of the six isomers of TMA.
The remaining isomers were numbered by Shulgin in the order of their progressive position of substitution, as abbreviated above. The values for relative human potencies of TMA-4, -5, and -6 compared to that of mescaline were evaluated to be 4, 10, and 10, respectively. TMA-3 was found to exhibit no psychotomimetic activity. The isomers, except TMA-3, have both stimulant and psychedelic effects, and their qualitative effects on mood alteration and sensory enhancement are similar to those of mescaline. Although their physical health risks and toxic doses in humans have never been clarified, mental health risks are assumed to be similar to those of other hallucinogens.

http://citeseerx.ist.psu.edu/viewdoc/download?doi=10.1.1.689.8613&rep=rep1&type=pdf
TMA-2:
Synthesised in 1947, TMA-2 is one of the numerous 'new synthetic drugs' with no legitimate therapeutic use that are described in Shulgin's Pikhal. TMA-2 can be produced from the active agent asarone, which is extracted from the rhizome of the plant Acorus calamus. The precursor asarone is widely used as an active principle in food flavourings. TMA-2 has the structural characteristics of amphetamines, which are associated with hallucinogenic and stimulant actions. It is an analogue that is very close to TMA but is 10 times more potent (20 mg of TMA-2 induces the same hallucinogenic effects as 200 mg of TMA). Therefore it carries potential risks common to TMA and other hallucinogenic substances (e.g. DOM, DOB)


https://www.emcdda.europa.eu/system/files/publications/322/Risk7_62957.pdf
TMA-2:
There are occasional references to the use of TMA-2 in early 1970's counter-culture publications, describing the drug as being rarely produced for sale and expensive in comparison to LSD. This suggests that there was only very limited human usage of TMA-2 between its invention and the publication of its synthesis and pharmacology in Shulgin's 1991 book PiHKAL. Since then it has been regarded as an oddball and a novelty in the psychedelics community and is only occasionally sought after intentionally. In terms of its subjective effects, it is known for its lack of classic psychedelic visuals compared to its parent compound (mescaline) and is known instead for its instead a unique stimulating body-high and altered headspace that ranges from euphoric to dysphoric variably.
Anecdotal reports suggest that TMA-2 is a highly unpredictable and dose-sensitive substance that can produce uncomfortable amounts of body load, nausea, overstimulation, and inconsistencies between experiences.
In modern times, TMA-2 is used as an obscure recreational drug and a occasional entheogen. It is rarely sold on the streets and is almost exclusively obtainable as a grey area research chemical through the use of online vendors.
| TMA-2 Duration: Trimethoxyamphetamine-2, a psychedelic amphetamine and stimulant first synthesised by Alexander Shulgin. An uncommon compound of similar activity to other psychedelic amphetamines. Short history of human use. | |||
| Route | Onset | Duration | After Effects |
|---|---|---|---|
| Tripsit Factsheets | |||

TMA-2 Basic Information: https://drugs.tripsit.me/tma-2 | |||
| All ROAs: | 20-120 minutes | 6-12 hours | Possibly hours |

| |||
| Effects: Euphoria, empathy, insight, brightened colour, Closed/Open eye visuals, enhanced tactile sensation, mental/physical stimulation, decreased appetite, pupil dilation, restlessness, change in perception, ego softening, sweating/chills, muscle tension, confusion, insomnia. | |||

https://www.bluelight.org/xf/threads/the-big-dandy-tma-series-thread.115050/

https://drugs-forum.com/tags/tma-2/

http://pihkal.info/read.php?domain=pk&id=157
#158 TMA-2:
http://pihkal.info/read.php?domain=pk&id=157
#159 TMA-3:
http://pihkal.info/read.php?domain=pk&id=157
#160 TMA-4:
http://pihkal.info/read.php?domain=pk&id=157
#161 TMA-5:
http://pihkal.info/read.php?domain=pk&id=157
#162 TMA-6:
http://pihkal.info/read.php?domain=pk&id=157


https://www.caymanchem.com/msdss/13887m.pdf


https://www.caymanchem.com/msdss/11144m.pdf
Trimethoxyamphetamine:
- A family of isomeric psychedelic hallucinogenic drugs.
- There exist six different TMAs that differ only in the position of the three methoxy groups: TMA, TMA-2, TMA-3, TMA-4, TMA-5, and TMA-6.
- The TMAs are analogs of the phenethylamine cactus alkaloid mescaline.
- First synthesized in 1947.
The most important TMA compound from a pharmacological standpoint is TMA-2, as this isomer has been much more widely used as a recreational drug and sold on the grey market as a so-called "research chemical"; TMA (sometimes referred to as "mescalamphetamine" or TMA-1) and TMA-6 have also been used in this way to a lesser extent. These three isomers are significantly more active as hallucinogenic drugs, and have consequently been placed onto the illegal drug schedules in some countries such as the Netherlands and Japan. The other three isomers TMA-3, TMA-4 and TMA-5 are not known to have been used as recreational drugs to any great extent, and remain obscure scientific curiosities.
The TMAs are substituted amphetamines, however, their action does not resemble that of the unsubstituted compound amphetamine, which is a stimulant and not a psychedelic. It is reported that some TMA's elicit a range of emotions ranging from sadness to empathy and euphoria.
ESCALINE
|
A positional isomer of TMA (3,4,5-trimethoxyamphetamine)
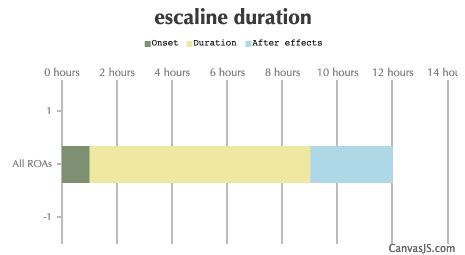
| Duration: First synthesised in the 50s, this uncommon drug was reexamined by David Nichols in the 1990s. It is an analogue of mescaline which is roughly six times more potent, and is thus a powerful psychedelic phenethylamine. Subjective effects may include stimulation and hallucinations. | |||
| Route | Onset | Duration | After Effects |
|---|---|---|---|
| Tripsit Factsheets | |||

Escaline Basic Information: http://drugs.tripsit.me/escaline | |||
| All ROAs: | 1.5-2 hours | 8-12 hours | 3-5 hours |
| |||
| Effects: Euphoria, empathy, insight, brightened colour, Closed/Open eye visuals, enhanced tactile sensation, mental/physical stimulation, decreased appetite, pupil dilation, restlessness, change in perception, ego softening, sweating/chills, muscle tension, confusion, insomnia. | |||
In comparison to mescaline, this compound has significantly more negative physical side effects, less insightful cognitive effects and less complex visual effects.
Tolerance and addiction potential
Escaline is not habit-forming, and the desire to use it can actually decrease with use. It is most often self-regulating. Tolerance to the effects of escaline is built almost immediately after ingestion. After that, it takes about 3 days for the tolerance to be reduced to half and 7 days to be back at baseline (in the absence of further consumption). Escaline presents cross-tolerance with all psychedelics, meaning that after the consumption of escaline all psychedelics will have a reduced effect.


https://psychonautwiki.org/wiki/Escaline
QUALITATIVE COMMENTS:
This is a powerful and complex intoxicant - I could not have coordinated any rational muscular activity. I could not walk; I could not tie my shoe-laces. There is analgesia and an incoordination that I cannot shake. My menstrual flow started a bit ahead of time, but it was light.
I felt that the body tensions outweighed the psychological and sensory rewards, in that I had a lot of dehydration and my sleep had a nightmare quality. This pretty much offset the few virtues that I felt I had obtained.
There is a quality of rational analysis and insight that is totally impressive. Many subtle factors in my life can be viewed with insight, and usefully dissected. I got into a deep discussion, but I was not argumentative or even defensive and I remained detached and kept a tone of cool impersonality. I had a good appetite. But I also had some tachycardia and muscular tension. There was unquestionable sensory enhancement, but without an intellectual component. Overall it was most pleasant.EXTENSIONS AND COMMENTARY:
In an isolated situation, there is easy fantasy, but little synthesis of external sensory inputs such as music or visual stimulae. A gradual decline brings the subject back to a restful baseline somewhere before the 12th hour. The following day is often seen as one of tiredness and low energy. An anonymous flyer appeared in the California drug community in 1984 stating an effective range to be 50 to 100 milligrams, but it described the drug as the sulfate. The above data all pertain to the hydrochloride salt.
 #72 E; Escaline:
#72 E; Escaline:
https://www.erowid.org/library/books_online/pihkal/pihkal072.shtml

https://erowid.org/experiences/subs/exp_TMA.shtml

https://erowid.org/experiences/subs/exp_Escaline.shtml

https://www.bluelight.org/xf/threads/the-big-dandy-escaline-thread.656624/

https://www.shroomery.org/forums/showflat.php/Number/18844491

https://www.reddit.com/search/?q=Escaline

https://www.quora.com/search?q=Escaline


https://www.caymanchem.com/msdss/14107m.pdf
Escaline:
- A positional isomer of TMA (3,4,5-trimethoxyamphetamine)
- A psychedelic drug
- First synthesized and reported in the scientific literature in 1954
- Mescaline analog
- The duration of action stated to be 8 - 12 hours
- 3,5-methoxy-4-ethoxyphenethylamine
- 2-(4-Ethoxy-3,5-dimethoxy-phenyl)-ethylamine
- 3,5-Dimethoxy-4-ethoxy-phenethylamine
- 2-(4-Ethoxy-3,5-dimethoxyphenyl)ethanamine
The effects of this and related mescaline analogues in humans were first described by Alexander Shulgin. In his book PiHKAL (Phenethylamines i Have Known And Loved), Shulgin lists the dosage range as 40 to 60 mg, consumed orally
FDA Approves Omeros’ YARTEMLEA® – First and Only Therapy Indicated for TA-TMA - The above button links to Coinbase. Yahoo Finance is not a broker-dealer or investment adviser and does not offer securities or cryptocurrencies for sale or facilitate trading. Coinbase pays us for ... Tuesday December 23, 2025 - finance.yahoo.com FDA Accepts Resubmission of BLA for Narsoplimab for Hematopoietic Stem Cell Transplant-Associated Thrombotic Microangiopathy (TA-TMA) and Assigns Late September PDUFA Date - The above button links to Coinbase. Yahoo Finance is not a broker-dealer or investment adviser and does not offer securities or cryptocurrencies for sale or facilitate trading. Coinbase pays us for ... Omeros Q1 earnings call highlights - Breakout Momentum Plays You Need to Know About Omeros (NASDAQ:OMER) reported first-quarter 2026 net income of $56.1 million, or $0.78 per share, as the company began commercial sales of YARTEMLEA, its ... Omeros stock jumps premarket after FDA approves first drug for rare transplant complication — retail sees big upside - ・The FDA approval clears the path for a U.S. launch in January 2026. ・Trial and expanded access data showed strong response rates and short-term survival in high-risk patients. ・Yartemlea becomes the ... First Drug for Deadly Transplant Complication Gets FDA's Blessing - The FDA has approved narsoplimab (Yartemlea) as the first treatment for hematopoietic stem cell transplant (HSCT)-associated thrombotic microangiopathy (TA-TMA) in adults and kids ages 2 years and up, ... FDA Approves Yartemlea, the First and Only Treatment for Transplant-Related Blood Vessel Damage - What Is Yartemlea, and Why Does It Matter? Yartemlea (narsoplimab-wuug) is a new medicine approved by the FDA for treating a rare and serious condition called transplant-associated thrombotic ... Unravel Biosciences Partners with TMA Precision Health to Advance Drug Treatment for Rett Syndrome into the Clinic - BOSTON, July 5, 2022 /PRNewswire/ -- Unravel Biosciences, Inc. ("Unravel"), a therapeutics company that spun out from the Wyss Institute for Biologically Inspired Engineering at Harvard University, ... TMA Infusion (10ml) (Chromium Picolinate) Drug Price and Information - For the above reasons, Medindia cannot be held liable for any action taken based on the information provided by its website. The purpose of this website is to provide initial guidance only and it does ... FDA Approves Omeros’ YARTEMLEA® – First and Only Therapy Indicated for TA-TMA - SEATTLE--(BUSINESS WIRE)-- Omeros Corporation (NASDAQ: OMER) today announced that the U.S. Food and Drug Administration (FDA) has approved YARTEMLEA® (narsoplimab-wuug) for the treatment of ... TMA Infusion (10ml) (Chromium Picolinate - Combination) Drug Price and Information - The information provided on this page is intended to serve as a comprehensive resource and should not be a substitute to professional medical advice. If you have concerns it is always best to speak ...
| ||
| Hallucinogens | Link to this page |






