[ Home ] [ Controlled Substances ] [ Opioids ]
BUTYRYL FENTANYL
|
Synthetic Opioid
World Health Organization 2016:
- Butyrfentanyl is a potent, short-acting mu opioid receptor agonist, and an analogue of fentanyl, differing by only one methyl group.
- It has no approved medical indications, but is being used for recreational purposes, with cases of fatal overdoses reported in Europe and the United States. The actual extent of abuse and mortality associated with butyrfentanyl is not known because it is not tested for in routine toxicological analyses.
- Typically used as a nasal spray, snorted as a white power, or injected intravenously, and doses range from 0.5 to 3 mg.
- The effects appear to be similar to the effects of fentanyl, including euphoria, altered mood states, drowsiness, and respiratory depression.
- Studies suggest that butyrfentanyl is about 30 times less potent than fentanyl
- Butyrfentanyl is being abused for its opioid effects.
- No accepted medical purpose has been identified.
| Duration: Potent short-acting opioid and fentanyl analogue. Often dispensed in a nasal spray. This is an highly potent drug - exercise extreme caution. May cause respiratory depression and death in overdose. NOTE: It's a potent drug, be careful. | |||
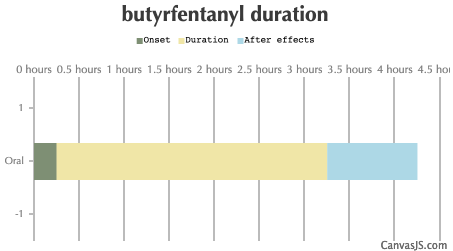
| Route | Onset | Duration | After Effects |
|---|---|---|---|
| Tripsit Factsheets | |||

Butyrfentanyl Basic Information: http://drugs.tripsit.me/butyrfentanyl | |||
| Oral: | 15-30 minutes | 3-4 hours | 1-4 hours |
| |||
| Avoid: Avoid all other CNS depressants. | |||
Aliases:
| |||

https://erowid.org/experiences/subs/exp_Butyrfentanyl.shtml
Butyrfentanyl:
- A synthetic opioid analgesic
- Potent
- Short-acting
- An analog of fentanyl with around one quarter of its potency
- Side effects are similar to those of fentanyl itself
- Butyrfentanyl binds to the opioid receptor
Butyrfentanyl has no current legitimate clinical applications; however, it is being sold as designer drug
Schedules of Controlled Substances: Temporary Placement of Butyryl Fentanyl and Beta-Hydroxythiofentanyl Into Schedule I - Barbara J. Boockholdt, Office of Diversion Control, Drug Enforcement Administration; Mailing Address: 8701 Morrissette Drive, Springfield, Virginia 22152; Telephone: (202) 598-6812. The Drug ... Tuesday March 22, 2016 - jurist.org Buffalo man charged after cocaine and butyryl fentanyl were seized in raid - A Buffalo man who was arrested on drug charges made an initial appearance Friday before U.S. Magistrate Michael J. Roemer and released on conditions, according to the U.S. Attorney's Office for the ... Fentanyl-like drug involved in New College overdose deaths - A powerful designer drug that has only recently gained the attention of authorities played a role in the accidental overdose deaths of two young men on New College's campus earlier this year, a ...
| ||
| Opioids | Link to this page |






