[ Home ] [ Controlled Substances ] [ Opioids ]
TILIDINE
|
How does Tilidine work?
The active ingredient tilidine influences the central nervous system by inhibiting the excitability of nerve cells: fewer pain signals are passed on to the brain. Tilidine is misused as a relaxation drug. It conveys a warm, calming feeling of wellbeing and relieves feelings of fear. It is also known to be used improperly in fights: the numbing effect is used to reduce pain. In addition, Tilidin increases the willingness to use violence and risk.
Because the pain reliever tilidine can develop a euphoric and disinhibiting effect when misused, it was used by drug addicts as a heroin substitute as early as the 1970s. Today Tilidin is partially played down in successful German rap songs, at the same time there is evidence that the Tilidin consumption by young adults has increased in recent years.
Dosage forms: Tilidine as drops or tablets:
- Germany Tilidine drops:
Tilidine is quickly effective in drop form, which is why it is used to treat acute painful conditions. A single administration can be sufficient, after which a switch is made to another pain reliever for further treatment. The analgesic effect of the Tilidine drops begins, depending on the manufacturer's instructions for use, after about ten to 15 minutes, after 25 to 50 minutes they have reached their maximum and the duration of action is about four to six hours. - Germany Tilidin-Retardtabletten:
The active ingredient tilidine is available in the form of prolonged-release tablets containing 50, 100, 150 or 200 mg tilidine hydrochloride. Due to the retardation (delay), the active ingredient is released more slowly in order to achieve long-lasting pain relief of up to twelve hours. Therefore, prolonged-release tablets must not be divided.
Due to the high potential for abuse, tilidine is now only available in combination with the active ingredient naloxone. Naloxone is an antagonist of tilidine: If tilidine is administered parenterally, e.g. injected with the aid of a syringe, the naloxone contained blocks the opiate receptors and in this way prevents the opioid from working. If, on the other hand, the pain reliever is taken orally as drops or tablets in appropriate doses, the so-called first-pass effect occurs: the naloxone is quickly broken down in the liver and tilidine is converted into nortilidine so that its effect can unfold.

https://tekdeeps.com/tilidine-effects-side-effects-dangers/
Tilidine is a low to medium potency opioid analgesic. It is metabolized to its active metabolites, nortilidine and bisnortilidine. Its analgesic activity is largely exerted through nortilidine which is a potent agonist at Mu opioid receptors.
 Tilidine:
Tilidine:
https://drugs.ncats.io/drug/GY33N31E9Y
Pharmacokinetics and Metabolism:
Tilidine is a prodrug, which, following oral administration, is converted to the active analgesic metabolite, nortilidine. The oral bioavailability of tilidine itself is of the order of 6 %. The half-life of elimination of nortilidine is of the order of 3 1/2 hours; peak plasma levels of nortilidine are achieved approximately 45 minutes following oral administration. Only 3 % of the oral dose is eliminated via the renal route in the form of nortilidine, and less than 0.1% as unchanged tilidine. The active metabolite nortilidine is eliminated by the hepatic route.


http://labeling.pfizer.com/ShowLabeling.aspx?id=1170
Tilidine is a prescription pain reliever here in Germany and a synthetically manufactured active ingredient. The active ingredient belongs to the group of opioids. By activating the opioid receptors, tilidine indirectly inhibits the pain-mediating neuronal systems in the body. Tilidine is converted to nortilidine in the liver. This process takes ten to 15 minutes after application.The duration of action of the pain reliever is about three to five hours.The pain reliever is addictive. Tilidine can be administered as an injection (syringe), tablet, prolonged release tablet or in the form of drops. The pain reliever has been around since the 1970s, but it wasn't until this decade that the pain reliever became really known due to its abuse in the German rap and hip hop scene. Couple of former dependent German music artists thematize Tilidine in their song lyrics:
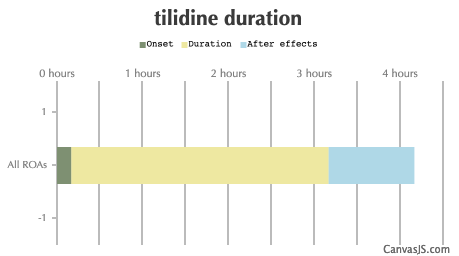
| Duration: Synthetic opioid, used mainly for treatment of moderate to severe pain. In some countries it's mixed with Naloxone to lower the abuse liability. Though in some only Tilidine. Roughly 0.2 - 1 on the potency scale to Morphine. (So 20mg's of Morphine to 100mg's Tilidine) | |||
| Route | Onset | Duration | After Effects |
|---|---|---|---|
| Tripsit Factsheets | |||

Tilidine Basic Information: http://drugs.tripsit.me/tilidine | |||
| All ROAs: | 10-20 minutes | 3-6 hours | 1-12 hours |
| |||
| Avoid: All other CNS depressants. | |||

https://erowid.org/experiences/subs/exp_Tilidine.shtml

https://www.reddit.com/search/?q=Tilidine

https://drugs-forum.com/threads/valoron-aka-tilidin.29034/
Tilidine:
Used mainly in Belgium, Bulgaria, Germany, Luxembourg, South Africa and Switzerland
- A synthetic opioid painkiller
- Used for the treatment of moderate to severe pain, both acute and chronic
- Its onset of pain relief after oral administration is about 10 - 15 minutes
- Peak relief from pain occurs about 25 - 50 minutes after oral administration
- The elimination half-life for nortilidine is 3-5 hours
- As well as its use as an analgesic, tilidine is also commonly used in Germany for treatment of restless legs syndrome
Adverse Effects:
Its most common adverse effects are transient nausea and vomiting, dizziness, drowsiness, fatigue, headache and nervousness; less commonly, nausea and vomiting (after repeated dosing), hallucinations, confusion, euphoria, tremor, hyperreflexia, clonus and increased sweating. Uncommonly, somnolence; rarely, diarrhoea and abdominal pain.In Germany, tilidine is available in a fixed combination with naloxone for oral administration (Valoron N and generics); the mixture of naloxone is claimed to lower the abuse liability of the opioid analgesic. This is so that if people take the medication orally (which is the way they are meant to) the opioid blocker, naloxone, has minimal effects on them but if they inject it the naloxone becomes bioavailable and hence antagonises the effects of the tilidine producing withdrawal effects.
Aoxing Pharmaceutical Starts producing Tilidine Hydrochloride Tablets - Aoxing Pharmaceutical Company, Inc. (NYSE MKT: AXN) disclosed that it commenced commercial production of Tilidine Hydrochloride tablets, which is a narcotic drug product. According to the company, ... Wednesday June 29, 2016 - finance.yahoo.com Aoxing Pharmaceutical Company, Inc. Announces Receipt Of License To Produce Tilidine HCL Tablet - JERSEY CITY, NJ / ACCESSWIRE / June 8, 2015 / Aoxing Pharmaceutical Company, Inc. (NYSE MKT: AXN) ("Aoxing Pharma"), a specialty pharmaceutical company focusing on research, development, manufacturing ...
| ||
| Opioids | Link to this page |




