[ Home ] [ Controlled Substances ] [ Depressants ]
TETRAZEPAM
|
Tetrazepam was the most widely prescribed muscle relaxant in Germany. After a series of studies, which have shown, that tetrazepam caused cutaneous adverse effects and toxic epidermal necrolysis, the drug was withdrawn from the market.
 Tetrazepam:
Tetrazepam:
https://drugs.ncats.io/drug/FO92091VP8
2013 Tetrazepam Suspension:
As a result of the risk of unpredictable, serious skin reactions identified, the CMDh position is that tetrazepam-containing medicines should no longer be used in the EU.
The review found that half of the reported reactions with tetrazepam are skin disorders, which are sometimes serious, life-threatening or fatal. Serious skin reactions include Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), erythema multiforme and DRESS syndrome. They are unpredictable and can occur at any stage during treatment, including after short-term treatment, and at recommended doses.

https://www.ema.europa.eu/en/news/recommendation-suspend-tetrazepam-containing-medicines-endorsed-cmdh


https://www.ema.europa.eu/en/documents/referral/tetrazepam-containing-medicines-article-107i-procedure-prac-recommends-suspension-tetrazepam_en.pdf
(2016) Tetrazepam Allergy:
Tetrazepam has been widely used in clinical praxis in many countries for over 50 years. As an agent belonging to the class of benzodiazepines, it has an immediate sedative, muscle relaxing, anxiolytic and anticonvulsant effect. According to the German drug prescription report for 2012, tetrazepam was the most commonly prescribed muscle relaxant after botulinum toxin. Common adverse reactions following systemic intake are of neurological and gastrointestinal nature, while cutaneous reactions to tetrazepam are very rare, with only a few cases reported in the literature. It has nevertheless to be assumed that only a minority of cases is reported. The accumulation of these adverse reactions in comparison with other muscle relaxing drugs led the European Commission to suspend marketing authorizations of tetrazepam-containing medicines across the European Union (EU) in 2013.


https://www.medicaljournals.se/acta/content_files/files/pdf/96/1/4443.pdf
On the clinical evidence leading to tetrazepam withdrawal:
In July 2013, the European Medicines Agency suspended the marketing authorization of tetrazepam across the European Union, due to serious cutaneous adverse drug reactions (ADR). Herein, we examine information described in PubMed and reported to the main Pharmacovigilance Databases (PhDB), related to ADR associated to tetrazepam. We tried to as certain described evidence on cutaneous ADR due to tetrazepam, which could have led to the end of commercialization of this drug that had been over 40 years in the market.
First, we conducted a search in MEDLINE and Cochrane (January 2015) about ADR due to tetrazepam, in peer-reviewed journals. Inclusion criteria were: studies performed on humans or tetrazepam-induced ADR case reports. Secondly, we collected data of spontaneous reporting of suspected ADR due to tetrazepam, from 1989 until December 2014, from the main PhDB: Spanish (FEDRA), French (BNPV) and American (FAERS).
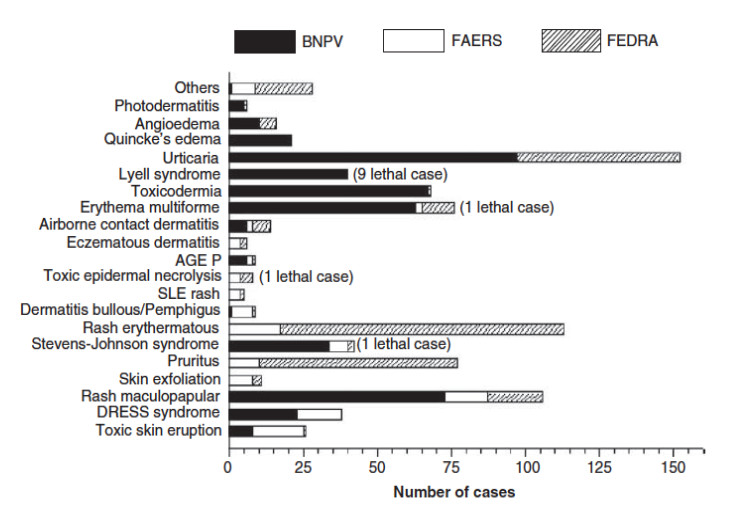
Our study reveals discrepancies in the information provided by these two different sources, both in the number of reported cases as well as in the kind of ADR reported. We stress the importance of a better knowledge communication between scientific literature and pharmacovigilance agencies, to prevent the use of marketed drugs with well-established side effects during long periods.


https://www.eahp.eu/sites/default/files/ps-031_0.pdf
- Allergic reactions can develop to tetrazepam and it is considered to be a potential allergen.
- Drug rash and drug-induced eosinophilia with systemic symptoms is a known complication of tetrazepam exposure. These hypersensitive allergic reactions can be of the delayed type.
- Drowsiness is a common side effect of tetrazepam.
- A reduction in muscle force can occur. Myasthenia gravis, a condition characterised by severe muscle weakness is another potential adverse effect from tetrazepam.
- Cardiovascular and respiratory adverse effects can occur with tetrazepam similar to other benzodiazepines.
- Prolonged use, as with all benzodiazepines, should be avoided, as tolerance occurs and there is a risk of benzodiazepine dependence and a benzodiazepine withdrawal syndrome after stopping or reducing dosage.
- Marketed under the following brand names, Clinoxan, Epsipam, Myolastan, Musaril, Relaxam and Spasmorelax.
Tetrazepam, like other benzodiazepines is a drug which is very frequently used in overdoses. These overdoses are often mixed overdoses, i.e. a mixture of other benzodiazepines or other drug classes with tetrazepam.
Small amounts of diazepam as well as the active metabolites of diazepam are produced from metabolism of tetrazepam. The metabolism of tetrazepam has led to false accusations of prisoners prescribed tetrazepam of taking illicit diazepam; this can lead to increased prison sentences for prisoners.
 Metabocard for Tetrazepam:
Metabocard for Tetrazepam:
https://hmdb.ca/metabolites/HMDB0042029
Contraindications:
- Acute narrow-angle glaucoma
- CNS depression or coma
- Myasthenia gravis
- Resp depression
- Coma
- Acute pulmonary insufficiency
- Sleep apnoea
- Severe hepatic impairment
- Porphyria
Special Precautions:
- Elderly or debilitated patients
- Pulmonary insufficiency
- Muscle weakness
- Organic brain changes
- Personality disorders
- Impaired liver or kidney function
- History of alcohol or drug addiction due to risk of dependence
- May precipitate suicide or aggressive behaviour
Adverse Reactions:
- Drowsiness
- Sedation
- Muscle weakness
- Ataxia
- Vertigo
- Headache
- Confusion
- Depression
- Slurred speech or dysarthria
- Changes in libido
- Tremor
- Visual disturbances
- Urinary retention or incontinence
- GI upsets
- Changes in salivation
- Amnesia
- Jaundice
- Blood disorders
- Hypersensitivity reactions
- Raised liver enzyme values
- Paradoxical excitation which may lead to hostility, aggression and disinhibition in some patients
Overdosage:
Impairment of consciousness, CNS depression, coma or paradoxical excitation. Symptomatic and supportive treatment. Activated charcoal may be given orally if within 1 hr of ingestion. Benzodiazepine antagonist, flumazenil, is rarely required and can be dangerous, especially in mixed overdoses involving TCA or in patients dependent on benzodiazepines.

https://www.mims.com/malaysia/drug/info/tetrazepam?mtype=generic
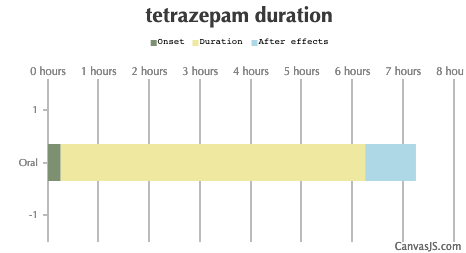
| Duration: A benzodiazepine that has most of the properties of more common benzodiazepines. Yet was taken off the market in 2013. Has been noted to be more euphoric than most others. | |||
| Route | Onset | Duration | After Effects |
|---|---|---|---|
| Tripsit Factsheets | |||

Tetrazepam Basic Information: http://drugs.tripsit.me/tetrazepam | |||
| Oral: | 15-35 minutes | 6-10 hours | 1-12 hours |
| |||
| Avoid: All other CNS depressants. | |||
| Chemical Safety Summary: |
|---|
| INCHEM |
| Main risks and target organs: |
| Central nervous system, causing depression of respiration and consciousness. |
| Summary of clinical effects: |
| Central nervous system (CNS) depression and coma, or paradoxical excitation, but deaths are rare when benzodiazepines are taken alone. Deep coma and other manifestations of severe CNS depression are rare. Sedation, somnolence, diplopia, dysarthria, ataxia and intellectual impairment are the most common adverse effects of benzodiazepines. Overdose in adults frequently involves co- ingestion of other CNS depressants, which act synergistically to increase toxicity. Elderly and very young children are more susceptible to the CNS depressant action. Intravenous administration of even therapeutic doses of benzodiazepines may produce apnoea and hypotension. Dependence may develop with regular use of benzodiazepines, even in therapeutic doses for short periods. If benzodiazepines are discontinued abruptly after regular use, withdrawal symptoms may develop. The amnesia produced by benzodiazepines can have medico-legal consequences. |
| Diagnosis: |
| The clinical diagnosis is based upon the history of benzodiazepine overdose and the presence of the clinical signs of benzodiazepine intoxication. Benzodiazepines can be detected or measured in blood and urine using standard analytical methods. This information may confirm the diagnosis but is not useful in the clinical management of the patient. A clinical response to flumazenil, a specific benzodiazepine antagonist, also confirms the diagnosis of benzodiazepine overdose, but administration of this drug is rarely justified. |
| First aid measures and management principles: |
| Most benzodiazepine poisonings require only clinical observation and supportive care. It should be remembered that benzodiazepine ingestions by adults commonly involve co- ingestion of other CNS depressants and other drugs. Activated charcoal normally provides adequate gastrointestinal decontamination. Gastric lavage is not routinely indicated. Emesis is contraindicated. The use of flumazenil is reserved for cases with severe respiratory or cardiovascular complications and should not replace the basic management of the airway and respiration. The routine use of flumazenil is contraindicated because of potential complications, including seizures. Renal and extracorporeal methods of enhanced elimination are not effective. |

https://www.reddit.com/search/?q=Tetrazepam

https://drugs-forum.com/threads/has-anyone-ever-tried-tetrazepam.22932/
It was formerly used mainly in Austria, France, Belgium, Germany and Spain to treat muscle spasm, anxiety disorders such as panic attacks, or more rarely to treat depression, premenstrual syndrome or agoraphobia.
Tetrazepam:
- A benzodiazepine derivative
- Anticonvulsant, anxiolytic, muscle relaxant and slightly hypnotic properties
- Has relatively little sedative effect at low doses while still producing useful muscle relaxation and anxiety relief.
- Tetrazepam is used therapeutically as a muscle relaxant.
- Allergic reactions to tetrazepam occasionally occur involving the skin.
- Drowsiness is a common side effect of tetrazepam.
- The benzodiazepine equivalent is approximately 100 mg of tetrazepam = 10 mg of diazepam.
- Tetrazepam, is rapidly absorbed after oral administration, within 45 mins and reaches peak plasma levels in less than 2 hours.
- An elimination half-life of approximately 15 hours
The European Commission has confirmed the suspension of the marketing authorisations for Tetrazepam in Europe because of cutaneous toxicity, effective August 2013
EU drugs agency recommends suspension of tetrazepam - LONDON, April 29 (Reuters) - EU drugs regulators have recommended suspending use of all tetrazepam-containing medicines following reports of serious skin reactions, the European Medicines Agency said ... Sunday April 28, 2013 - reuters.com European warning on Tetrazepam - SKIN reactions caused by the drug Tetrazepam, whose best-known brand name is Myolastan, have caused the European Medicines Agency advising market suspension. Tetrazepam, whose best-known brand name is ...
| ||
| Depressants | Link to this page |





